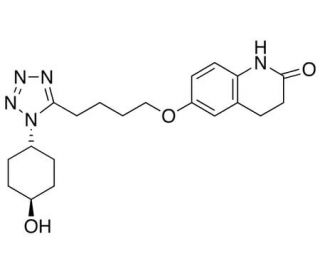
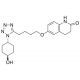
4′-trans-Hydroxy Cilostazol (CAS 87153-04-6)
See product citations (1)
QUICK LINKS
ZDHHC3 inhibitors are a class of chemical compounds designed to specifically target and inhibit the activity of ZDHHC3, a protein encoded by the ZDHHC3 gene. ZDHHC3, also known as zinc finger DHHC-type palmitoyltransferase 3, is an enzyme that plays a critical role in the process of protein palmitoylation. Palmitoylation is a post-translational modification where a palmitoyl group (a type of fatty acid) is covalently attached to cysteine residues on proteins, which influences their membrane association, trafficking, stability, and function. ZDHHC3 is one of several DHHC-type palmitoyltransferases, characterized by the presence of a DHHC (Asp-His-His-Cys) cysteine-rich domain, which is essential for its enzymatic activity. Structurally, ZDHHC3 inhibitors can vary widely, including small molecules, peptides, or other biomolecules engineered to interact specifically with the catalytic site or regulatory domains of the ZDHHC3 protein. These inhibitors typically bind to the DHHC domain or other critical regions, blocking the enzyme′s ability to transfer palmitoyl groups to substrate proteins. By inhibiting ZDHHC3, these compounds can disrupt the palmitoylation process, leading to changes in the localization, stability, and function of target proteins. The study of ZDHHC3 inhibitors is crucial for understanding the regulatory mechanisms of protein palmitoylation and its impact on cellular signaling and membrane dynamics. Research into these inhibitors provides valuable insights into the broader implications of palmitoylation on protein function, including how alterations in this modification can affect cellular processes such as signal transduction, vesicular trafficking, and protein-protein interactions. By exploring the effects of ZDHHC3 inhibition, scientists can gain a deeper understanding of the role of palmitoylation in maintaining cellular homeostasis and the functional organization of the cell.
4′-trans-Hydroxy Cilostazol (CAS 87153-04-6) References
- The quantitative determination of cilostazol and its four metabolites in human liver microsomal incubation mixtures by high-performance liquid chromatography. | Tata, PN., et al. 1998. J Pharm Biomed Anal. 18: 441-51. PMID: 10096838
- Simultaneous quantitative determination of cilostazol and its metabolites in human plasma by high-performance liquid chromatography. | Fu, CJ., et al. 1999. J Chromatogr B Biomed Sci Appl. 728: 251-62. PMID: 10406210
- Method for the quantitative analysis of cilostazol and its metabolites in human plasma using LC/MS/MS. | Bramer, SL., et al. 2001. J Pharm Biomed Anal. 26: 637-50. PMID: 11516916
- Inhibition of lipopolysaccharide-induced apoptosis by cilostazol in human umbilical vein endothelial cells. | Kim, KY., et al. 2002. J Pharmacol Exp Ther. 300: 709-15. PMID: 11805237
- Cilostazol to overcome high on-treatment platelet reactivity in korean patients treated with clopidogrel and calcium-channel blocker. | Jeong, YH., et al. 2011. Circ J. 75: 2534-6. PMID: 21970840
- K-134, a phosphodiesterase 3 inhibitor, prevents brain damage by inhibiting thrombus formation in a rat cerebral infarction model. | Yoshida, H., et al. 2012. PLoS One. 7: e46432. PMID: 23110051
- Aspirin and probenecid inhibit organic anion transporter 3-mediated renal uptake of cilostazol and probenecid induces metabolism of cilostazol in the rat. | Wang, C., et al. 2014. Drug Metab Dispos. 42: 996-1007. PMID: 24692216
- Effects of cilostazol on the pharmacokinetics of carvedilol after oral and intravenous administration in rats. | Lim, TH., et al. 2015. J Physiol Pharmacol. 66: 591-7. PMID: 26348083
- Organic Anion-Transporting Polypeptide and Efflux Transporter-Mediated Hepatic Uptake and Biliary Excretion of Cilostazol and Its Metabolites in Rats and Humans. | Wang, C., et al. 2017. J Pharm Sci. 106: 2515-2523. PMID: 28535976
- Pharmacokinetic modeling analysis of cilostazol and its active metabolites (OPC-13015 and OPC-13213) after multiple oral doses of cilostazol in healthy Korean volunteers. | Cui, A., et al. 2020. Xenobiotica. 50: 288-296. PMID: 31181990
- Conversion from cilostazol to OPC-13015 linked to mitigation of cognitive impairment. | Saito, S., et al. 2021. Alzheimers Dement (N Y). 7: e12182. PMID: 34095441
- Impact of Cilostazol Pharmacokinetics on the Development of Cardiovascular Side Effects in Patients with Cerebral Infarction. | Yokoyama, T., et al. 2021. Biol Pharm Bull. 44: 1767-1774. PMID: 34719653
- Post-stroke treatment with K-134, a phosphodiesterase 3 inhibitor, improves stroke outcomes in the stroke-prone spontaneously hypertensive rat model-A comparative evaluation of antiplatelet drugs. | Yoshida, H., et al. 2022. J Pharmacol Sci. 148: 229-237. PMID: 35063138
- The metabolism of a new antithrombotic and vasodilating agent, cilostazol, in rat, dog and man. | Akiyama, H., et al. 1985. Arzneimittelforschung. 35: 1133-40. PMID: 4074424
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4′-trans-Hydroxy Cilostazol, 5 mg | sc-210206 | 5 mg | $401.00 |
