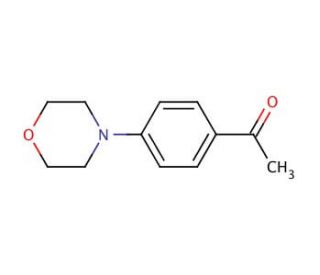
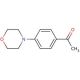
4′-Morpholinoacetophenone (CAS 39910-98-0)
QUICK LINKS
4′-Morpholinoacetophenone is a chemical compound that functions as a reagent in organic synthesis. It acts as a key intermediate in the preparation of various organic compounds, particularly those with agrochemical applications. Its mechanism of action involves participating in nucleophilic substitution reactions, where the morpholino group serves as a nucleophile, facilitating the formation of new carbon-carbon or carbon-heteroatom bonds. 4′-Morpholinoacetophenone′s ability to undergo selective functionalization at the para position of the phenyl ring, making it a versatile building block in the synthesis of complex molecules. 4′-Morpholinoacetophenone plays a role in the construction of diverse chemical structures, contributing to the advancement of methodologies and the discovery of novel compounds with potential biological activities. Its reactivity and selectivity make it useful for chemists working on the design and synthesis of new chemical entities.
4′-Morpholinoacetophenone (CAS 39910-98-0) References
- A New Potent and Selective Monoamine Oxidase-B Inhibitor with Extended Conjugation in a Chalcone Framework: 1-[4-(Morpholin-4-yl)phenyl]-5-phenylpenta-2,4-dien-1-one. | Maliyakkal, N., et al. 2020. ChemMedChem. 15: 1629-1633. PMID: 32583952
- Thiazolyl-pyrazoline derivatives: In vitro and in silico evaluation as potential acetylcholinesterase and carbonic anhydrase inhibitors. | Sever, B., et al. 2020. Int J Biol Macromol. 163: 1970-1988. PMID: 32931834
- Identification of a new class of potent aldose reductase inhibitors: Design, microwave-assisted synthesis, in vitro and in silico evaluation of 2-pyrazolines. | Sever, B., et al. 2021. Chem Biol Interact. 345: 109576. PMID: 34252406
- Syntheses and anti-inflammatory activities of O-acyloximes. II. | Katagi, T., et al. 1996. Chem Pharm Bull (Tokyo). 44: 145-9. PMID: 8582034
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4′-Morpholinoacetophenone, 5 g | sc-233123 | 5 g | $41.00 |
