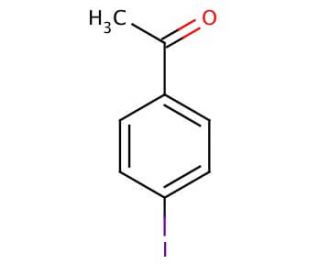
4′-Iodoacetophenone (CAS 13329-40-3)
QUICK LINKS
4′-Iodoacetophenone is a compound of interest in organic chemistry research due to its utility as a precursor in various synthetic pathways. Its iodo and carbonyl functional groups make it a versatile reagent for cross-coupling reactions, such as the Suzuki or Heck reactions, allowing for the construction of more complex organic systems. Researchers value it for the introduction of iodine into aromatic rings, which can then be used in further functionalization steps, such as halogen exchange or oxidative addition in palladium-catalyzed processes. The acetophenone moiety of the molecule also plays a role in studies of aromatic ketones, where it can undergo enolization, reduction, or serve as a building block for heterocyclic compounds. The ability to modify the phenyl ring through electrophilic aromatic substitution makes 4′-Iodoacetophenone a pivotal compound in the design and synthesis of new materials, dyes, and other chemicals, providing a pathway to explore novel chemical properties and reactions.
4′-Iodoacetophenone (CAS 13329-40-3) References
- Bacterial degradation of ring-chlorinated acetophenones. | Higson, FK. and Focht, DD. 1990. Appl Environ Microbiol. 56: 3678-85. PMID: 16348372
- Polyionic polymers--heterogeneous media for metal nanoparticles as catalyst in Suzuki-Miyaura and Heck-Mizoroki reactions under flow conditions. | Mennecke, K. and Kirschning, A. 2009. Beilstein J Org Chem. 5: 21. PMID: 19590749
- A Highly Efficient and Reusable Palladium(II)/Cationic 2,2'-Bipyridyl-Catalyzed Stille Coupling in Water. | Wu, WY., et al. 2016. Molecules. 21: PMID: 27617999
- Synergistic palladium/enamine catalysis for asymmetric hydrocarbon functionalization of unactivated alkenes with ketones. | Wei, C., et al. 2019. Org Biomol Chem. 17: 6607-6611. PMID: 31237304
- Polyethyleneimine-Modified Polymer as an Efficient Palladium Scavenger and Effective Catalyst Support for a Functional Heterogeneous Palladium Catalyst. | Yamada, T., et al. 2019. ACS Omega. 4: 10243-10251. PMID: 31460116
- Synthesis, X-ray Characterization and Density Functional Theory (DFT) Studies of Two Polymorphs of the α,α,α,α, Isomer of Tetra-p-Iodophenyl Tetramethyl Calix[4]pyrrole: On the Importance of Halogen Bonds. | Dăbuleanu, D., et al. 2020. Molecules. 25: PMID: 31936802
- Synthesis of β-Phenethylamines via Ni/Photoredox Cross-Electrophile Coupling of Aliphatic Aziridines and Aryl Iodides. | Steiman, TJ., et al. 2020. J Am Chem Soc. 142: 7598-7605. PMID: 32250602
- Structure-Guided Optimization of Replication Protein A (RPA)-DNA Interaction Inhibitors. | Gavande, NS., et al. 2020. ACS Med Chem Lett. 11: 1118-1124. PMID: 32550990
- Gold(I)-Catalyzed Reactivity of Furan-ynes with N-Oxides: Synthesis of Substituted Dihydropyridinones and Pyranones. | Nejrotti, S., et al. 2021. J Org Chem. 86: 8295-8307. PMID: 34100288
- C4-arylation and domino C4-arylation/3,2-carbonyl migration of indoles by tuning Pd catalytic modes: Pd(i)-Pd(ii) catalysis vs. Pd(ii) catalysis. | Cheng, Y., et al. 2021. Chem Sci. 12: 3216-3225. PMID: 34164090
- Zn-Nx sites on N-doped carbon for aerobic oxidative cleavage and esterification of C(CO)-C bonds. | Xie, C., et al. 2021. Nat Commun. 12: 4823. PMID: 34376654
- Leveraging of Sulfur Anions in Photoinduced Molecular Transformations. | Li, H., et al. 2021. JACS Au. 1: 2121-2129. PMID: 34977884
- Mechanically Robust Hybrid Gel Beads Loaded with 'Naked' Palladium Nanoparticles as Efficient, Reusable, and Sustainable Catalysts for the Suzuki-Miyaura Reaction. | Albino, M., et al. 2023. ACS Sustain Chem Eng. 11: 1678-1689. PMID: 36778525
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4′-Iodoacetophenone, 5 g | sc-252232 | 5 g | $14.00 | |||
4′-Iodoacetophenone, 25 g | sc-252232A | 25 g | $28.00 | |||
4′-Iodoacetophenone, 100 g | sc-252232B | 100 g | $74.00 |
