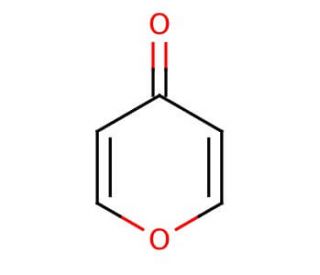
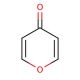
4H-Pyran-4-one (CAS 108-97-4)
QUICK LINKS
4H-Pyran-4-one is a chemical compound that functions as a versatile intermediate in organic synthesis. It serves as a key building block for the preparation of various heterocyclic compounds, including agrochemicals, and materials. Its mode of action involves participating in a range of chemical reactions, such as cycloaddition, oxidation, and substitution, to form complex molecular structures. Reactivity allows for the modification of its structure, enabling the creation of diverse derivatives with unique properties. In the development, 4H-Pyran-4-one plays a role in the synthesis of novel compounds for further investigation and potential application in various fields. Its ability to undergo multiple transformations makes it useful for the exploration of new chemical entities and the development of innovative materials.
4H-Pyran-4-one (CAS 108-97-4) References
- Use of semiempirical methods for the prediction of equilibrium geometries and ionization potentials of 4h-pyran-4-one and sulphur analogues | Á Somogyi, Z Dinya, A Császár, P Császár - Journal of Molecular Structure …, 1985 - Elsevier. November 1985,. Journal of Molecular Structure: THEOCHEM. Volume 133,: Pages 95-104.
- Theoretical force fields and vibrational spectra of 4H-pyran-4-one by CNDO/2 and MINDO/3 force methods | Á Somogyi, P Császár, Z Dinya, AG Császár - Journal of Molecular …, 1987 - Elsevier. May 1987,. Journal of Molecular Structure: THEOCHEM. Volume 151,: Pages 29-37.
- The reaction of ethyl 4H-pyran-4-one-2-carboxylate with 1,2-diaminobenzene | and Diether G. Markees. September/October 1990. Journal of Heterocyclic Chemistry. Volume27, Issue6: Pages 1837-1838.
- Synthesis of New 1,2,3-Triazole Derivatives Possessing 4H-Pyran-4-one Moiety by 1,3-Dipolar Cycloaddition Reaction of Azidomethyl Phenylpyrone with Various Alkynes | and Mahnaz Saraei, Bagher Eftekhari-Sis, Massomeh Faramarzi, Roshanak Hossienzadeh. September 2014. Journal of Heterocyclic Chemistry. Volume51, Issue5: Pages 1500-1503.
- A convenient synthesis of 5-arylamino-4H-pyran-4-ones using palladium-catalyzed amination | J Farard, C Logé, B Pfeiffer, B Lesur, M Duflos - Tetrahedron Letters, 2009 - Elsevier. 14 October 2009,. Tetrahedron Letters. Volume 50, Issue 41,: Pages 5729-5732.
- Facile synthesis of caerulomycin E by the formation of 2,2′-bipyridine core via a 2-pyridyl substituted 4H-pyran-4-one. Formal synthesis of caerulomycin A | DN Bobrov, VI Tyvorskii - Tetrahedron, 2010 - Elsevier. 17 July 2010,. Tetrahedron. Volume 66, Issue 29,: Pages 5432-5434.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4H-Pyran-4-one, 250 mg | sc-239012 | 250 mg | $39.00 | |||
4H-Pyran-4-one, 1 g | sc-239012A | 1 g | $123.00 |
