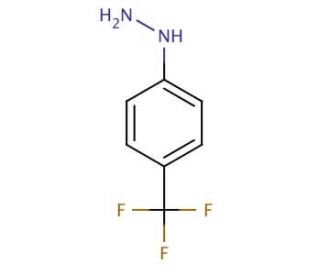
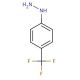
4-(Trifluoromethyl)phenylhydrazine (CAS 368-90-1)
QUICK LINKS
4-(Trifluoromethyl)phenylhydrazine serves as an intermediate in the synthesis of a variety of nitrogen-containing heterocycles, which are of interest in materials science for their potential electronic and photonic properties. Researchers utilize this compound to introduce the trifluoromethyl functional group into target molecules, aiming to exploit its electron-withdrawing capabilities to modulate the electronic character of these molecules. In the field of organic electronics, the introduction of such fluorinated groups may act to significantly alter the charge transport properties of organic semiconductors, which is for the development of high-performance optoelectronic devices. The hydrazine moiety of 4-(Trifluoromethyl)phenylhydrazine is also actively studied for its role in the synthesis of agrochemicals, where it can contribute to the creation of compounds with a range of potential activity. Furthermore, this compound is explored in the design of novel organic dyes and pigments, where the stability and color properties of the final product can be finely tuned for specific applications.
4-(Trifluoromethyl)phenylhydrazine (CAS 368-90-1) References
- 2-Substituted paullones: CDK1/cyclin B-inhibiting property and in vitro antiproliferative activity. | Kunick, C., et al. 2000. Bioorg Med Chem Lett. 10: 567-9. PMID: 10741555
- An efficient synthesis of achiral and chiral 1,2,4-triazolium salts: bench stable precursors for N-heterocyclic carbenes. | Kerr, MS., et al. 2005. J Org Chem. 70: 5725-8. PMID: 15989360
- Synthesis and in vitro antitumor and antimicrobial activity of some 2,3-diaryl-7-methyl-4,5,6,7-tetrahydroindazole and 3,3a,4,5,6,7-hexahydroindazole derivatives. | Faidallah, HM., et al. 2013. J Enzyme Inhib Med Chem. 28: 495-508. PMID: 22329488
- Synthesis of Unsymmetrical 2,6-Diarylanilines by Palladium-Catalyzed C-H Bond Functionalization Methodology. | Kwak, SH., et al. 2018. J Org Chem. 83: 5844-5850. PMID: 29737848
- Tuning a pyrazoline-based fluorogenic reagent, 3-naphthyl-1-(4-trifluoromethyl)-5-(4-carboxy phenyl)-2-pyrazoline for sensitive precolumn derivatization of aliphatic alcohols and trace analysis by RP-HPLC with fluorescence detection. | Al-Sabahi, A., et al. 2021. Biomed Chromatogr. 35: e5134. PMID: 33942331
- Conditions to Control Furan Ring Opening during Furfuryl Alcohol Polymerization. | Quinquet, L., et al. 2022. Molecules. 27: PMID: 35630693
- Growth vector elaboration of fragments: regioselective functionalization of 5-hydroxy-6-azaindazole and 3-hydroxy-2,6-naphthyridine. | Eliandro da Silva Júnior, P., et al. 2022. Org Biomol Chem. 20: 7483-7490. PMID: 36102876
- Modular Synthesis of Phosphino Hydrazones and Their Use as Ligands in a Palladium-Catalysed Cu-Free Sonogashira Cross-Coupling Reaction. | Baweja, S., et al. 2023. Chempluschem. e202300163. PMID: 37155325
- Quantitative Low-Field 19F NMR Analysis of Carbonyl Groups in Pyrolysis Oils. | Tang, B., et al. 2023. ChemSusChem. e202300625. PMID: 37318880
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-(Trifluoromethyl)phenylhydrazine, 5 g | sc-232327 | 5 g | $86.00 |
