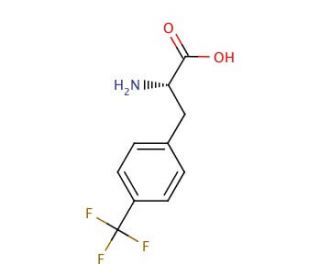
4-(Trifluoromethyl)-L-phenylalanine (CAS 114926-38-4)
QUICK LINKS
4-(Trifluoromethyl)-L-phenylalanine is a compound that functions as a non-natural amino acid in peptide synthesis. It is used to introduce a trifluoromethyl group into peptides, which can alter their physicochemical properties and biological activity. The mechanism of action of 4-(Trifluoromethyl)-L-phenylalanine involves its incorporation into the peptide sequence during solid-phase peptide synthesis. This non-natural amino acid can modulate the peptide′s conformation and stability, as well as its interactions with biological targets. At the molecular level, 4-(Trifluoromethyl)-L-phenylalanine may influence the peptide′s binding affinity, enzymatic activity, or cellular uptake, depending on the specific sequence and context. Its unique chemical structure allows for the exploration of structure-activity relationships and the development of peptide-based probes or potential biologically active compounds.
4-(Trifluoromethyl)-L-phenylalanine (CAS 114926-38-4) References
- The introduction of fluorine atoms or trifluoromethyl groups in short cationic peptides enhances their antimicrobial activity. | Giménez, D., et al. 2006. Bioorg Med Chem. 14: 6971-8. PMID: 16809042
- Glycosaminoglycan Binding and Non-Endocytic Membrane Translocation of Cell-Permeable Octaarginine Monitored by Real-Time In-Cell NMR Spectroscopy. | Takechi-Haraya, Y., et al. 2017. Pharmaceuticals (Basel). 10: PMID: 28420127
- Current Understanding of Physicochemical Mechanisms for Cell Membrane Penetration of Arginine-rich Cell Penetrating Peptides: Role of Glycosaminoglycan Interactions. | Takechi-Haraya, Y. and Saito, H. 2018. Curr Protein Pept Sci. 19: 623-630. PMID: 29332576
- Solution NMR to Quantify Mobility in Membranes: Diffusion, Protrusion, and Drug Transport Processes. | Okamura, E. 2019. Chem Pharm Bull (Tokyo). 67: 308-315. PMID: 30930433
- Design and Combinatorial Development of Shield-1 Peptide Mimetics Binding to Destabilized FKBP12. | Madsen, D., et al. 2020. ACS Comb Sci. 22: 156-164. PMID: 32027120
- Modulating co-translational protein folding by rational design and ribosome engineering. | Ahn, M., et al. 2022. Nat Commun. 13: 4243. PMID: 35869078
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-(Trifluoromethyl)-L-phenylalanine, 1 g | sc-226438 | 1 g | $206.00 |
