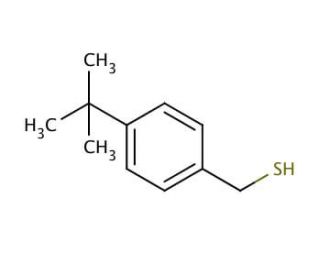
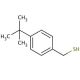
4-tert-Butylbenzyl mercaptan (CAS 49543-63-7)
QUICK LINKS
4-tert-Butylbenzyl mercaptan is a very useful reactant. For example, it can be used for stereoselective thiourea-catalyzed sulfa-Michael addition, nucleophilic substitution reactions, urea-catalyzed trans-thioesterification, sc-catalyzed Michael addition, and copper-catalyzed decarboxylative cross-coupling. 4-tert-Butylbenzyl mercaptan (4-TBB) is a versatile volatile aliphatic mercaptan, frequently employed in synthesizing other mercaptans and playing a role as a reagent in organic synthesis. Beyond its industrial applications, this compound finds its way into the food and beverage industry as a flavor and fragrance additive, thanks to its colorless liquid form and a distinctive sulfurous aroma. In the realm of scientific research, 4-tert-Butylbenzyl mercaptan has garnered attention for its broad range of applications. It has proven valuable in the synthesis of various mercaptans, and its role as a reagent extends into biochemical and physiological studies. Moreover, this compound serves as an excellent model for examining how mercaptans impact the environment. Within the food and beverage industry, 4-tert-Butylbenzyl mercaptan is believed to act as both an odorant and a potentiator, enhancing the flavors of specific foods and drinks. Its influence in augmenting taste experiences makes it a sought-after ingredient. Additionally, 4-tert-Butylbenzyl mercaptan has demonstrated its inhibitory effects on certain enzymes, including lipoxygenase, in the metabolism of certain compounds. This inhibition reveals further potential applications in various fields, making this compound an intriguing subject of scientific interest. 4-tert-Butylbenzyl mercaptan is an important aliphatic mercaptan with a multifaceted presence in organic synthesis, food and beverage flavoring, and scientific research, hinting at its valuable contributions across diverse domains.
4-tert-Butylbenzyl mercaptan (CAS 49543-63-7) References
- Reaction of triphenylphosphine with phenylethanethiolate-protected Au38 nanoparticles. | Wang, W. and Murray, RW. 2005. Langmuir. 21: 7015-22. PMID: 16008417
- Arylthiolate-protected silver quantum dots. | Branham, MR., et al. 2006. Langmuir. 22: 11376-83. PMID: 17154628
- Synthesis and biological evaluation of pyridazinone analogues as potential cardiac positron emission tomography tracers. | Purohit, A., et al. 2008. J Med Chem. 51: 2954-70. PMID: 18422306
- Discovery and optimization of aminopyrimidinones as potent and state-dependent Nav1.7 antagonists. | Nguyen, HN., et al. 2012. Bioorg Med Chem Lett. 22: 1055-60. PMID: 22209205
- The dynamic kinetic resolution of azlactones with thiol nucleophiles catalyzed by arylated, deoxygenated cinchona alkaloids. | Rodríguez-Docampo, Z., et al. 2012. J Org Chem. 77: 2407-14. PMID: 22353170
- New Protocols for the Synthesis of Stable Ag and Au Nanocluster Molecules. | Udayabhaskararao, T. and Pradeep, T. 2013. J Phys Chem Lett. 4: 1553-64. PMID: 26282314
- Ag50(Dppm)6(SR)30 and Its Homologue AuxAg50-x(Dppm)6(SR)30 Alloy Nanocluster: Seeded Growth, Structure Determination, and Differences in Properties. | Du, W., et al. 2017. J Am Chem Soc. 139: 1618-1624. PMID: 28111946
- Sharp Transition from Nonmetallic Au246 to Metallic Au279 with Nascent Surface Plasmon Resonance. | Higaki, T., et al. 2018. J Am Chem Soc. 140: 5691-5695. PMID: 29658712
- Sensitive Assays by Nucleophile-Induced Rearrangement of Photoactivated Diarylethenes. | Fredrich, S., et al. 2018. J Am Chem Soc. 140: 6432-6440. PMID: 29756777
- A thirty-fold photoluminescence enhancement induced by secondary ligands in monolayer protected silver clusters. | Khatun, E., et al. 2018. Nanoscale. 10: 20033-20042. PMID: 30351319
- Approaching Materials with Atomic Precision Using Supramolecular Cluster Assemblies. | Chakraborty, P., et al. 2019. Acc Chem Res. 52: 2-11. PMID: 30507167
- Formation of an NIR-emitting Ag34S3SBB20(CF3COO)62+ cluster from a hydride-protected silver cluster. | Manju, CK., et al. 2019. Dalton Trans. 48: 8664-8670. PMID: 31135003
- Oxidative cleavage and ammoxidation of organosulfur compounds via synergistic Co-Nx sites and Co nanoparticles catalysis. | Luo, H., et al. 2023. Nat Commun. 14: 2981. PMID: 37221164
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-tert-Butylbenzyl mercaptan, 25 g | sc-232996 | 25 g | $31.00 |
