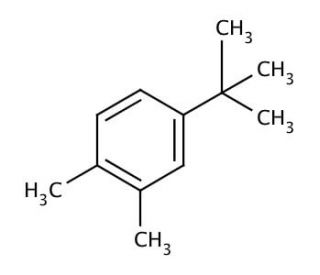
4-tert-Butyl-o-xylene (CAS 7397-06-0)
QUICK LINKS
4-tert-Butyl-o-xylene is a chemically modified version of xylene, specifically an o-xylene where a tert-butyl group is added to the fourth position of the aromatic benzene ring. This structural modification significantly alters the physical and chemical properties of the original molecule, enhancing its stability and modifying its reactivity. In research, 4-tert-Butyl-o-xylene is particularly valued for its applications in materials science and organic synthesis, where its bulky tert-butyl group plays a crucial role. The introduction of the tert-butyl group increases the steric hindrance around the benzene ring, which can inhibit unwanted side reactions and direct the course of chemical reactions to desired products. This makes it an excellent candidate for use in the synthesis of more complex aromatic compounds where precise control over reaction pathways is necessary. Additionally, the modified electronic properties of the benzene ring due to the presence of the tert-butyl group affect the molecule′s interaction with various reagents and catalysts, offering unique insights into the mechanisms of aromatic substitution reactions. Research involving 4-tert-Butyl-o-xylene contributes to a deeper understanding of the influence of substituents on the reactivity and properties of benzene derivatives, which is critical for the development of new synthetic materials and chemicals with tailored properties.
4-tert-Butyl-o-xylene (CAS 7397-06-0) References
- Organic reactions catalyzed by solid superacids. 10. Perfluorinated sulfonic acid resin (Nafion-H) catalyzed ring closure reaction of 2,2'-diaminobiphenyls. A preparative route to carbazoles | Yamato, Takehiko, et al. 1991. The Journal of Organic Chemistry. 56: 6248-6250.
- Friedel–Crafts green alkylation of xylenes with tert-butanol over mesoporous superacid UDCaT-5 | Yadav, Ganapati D., and Shashikant B. Kamble. 2012. Chemical Engineering Research and Design. 90: 1322-1334.
- Iodinated zinc phthalocyanine – The novel visible-light activated photosensitizer for efficient generation of singlet oxygen | Cyza, Małgorzata, et al. 2018. Journal of Photochemistry and Photobiology A. 358: 265-273.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-tert-Butyl-o-xylene, 5 ml | sc-232989 | 5 ml | $32.00 |
