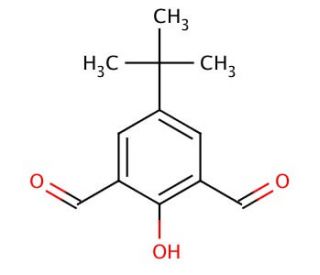
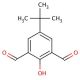
Molecular structure of 4-tert-Butyl-2,6-diformylphenol, CAS Number: 84501-28-0
4-tert-Butyl-2,6-diformylphenol (CAS 84501-28-0)
CAS Number:
84501-28-0
Molecular Weight:
206.24
Molecular Formula:
C12H14O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-tert-Butyl-2,6-diformylphenol is an organic compound featuring an aldehyde group, a tert-butyl group, and a hydroxy group connected to an isophthalic acid fragment. It presents itself as a colorless crystalline solid. It assumes a pivotal role as an intermediate in the synthesis of diverse organic compounds. Furthermore, it finds application in polymer synthesis and the creation of other chemical substances. While the precise mechanism of action of 4-tert-Butyl-2,6-diformylphenol remains unclear, it is believed to function as an inhibitor of the enzyme cyclooxygenase-2 (COX-2).
4-tert-Butyl-2,6-diformylphenol (CAS 84501-28-0) References
- Controlling the macrocycle size by the stoichiometry of the applied template ion. | Sarnicka, A., et al. 2012. Chem Commun (Camb). 48: 2237-9. PMID: 22252253
- Structural, electrochemical, and spectroscopic investigation of acetate bridged dinuclear tetrakis-Schiff base macrocycles of Mn and Zn. | Kal, S., et al. 2013. Inorg Chem. 52: 13963-73. PMID: 24262031
- Electrocatalytic proton reduction by a dicobalt tetrakis-Schiff base macrocycle in nonaqueous electrolyte. | Kal, S., et al. 2014. Inorg Chem. 53: 7137-45. PMID: 24963755
- Transition metal(II) complexes of a novel symmetrical benzothiazole-based ligand: synthesis, spectral/structural characterization and fluorescence properties. | Gulcan, M., et al. 2014. J Fluoresc. 24: 1679-86. PMID: 25212791
- Tetranuclear and Pentanuclear Compounds of the Rare-Earth Metals: Synthesis and Magnetism. | Yadav, M., et al. 2015. Inorg Chem. 54: 7846-56. PMID: 26218434
- Trinuclear Cage-Like Zn(II) Macrocyclic Complexes: Enantiomeric Recognition and Gas Adsorption Properties. | Janczak, J., et al. 2016. Chemistry. 22: 598-609. PMID: 26642975
- Zn2+ -Ion Sensing by Fluorescent Schiff Base Calix[4]arene Macrocycles. | Ullmann, S., et al. 2017. Chemistry. 23: 3824-3827. PMID: 28195665
- Binuclear Double-Stranded Helicates and Their Catalytic Applications in Desymmetrization of Mesodiols. | Chinnaraja, E., et al. 2019. Inorg Chem. 58: 4465-4479. PMID: 30929448
- Chiral metal-organic cages used as stationary phase for enantioseparations in capillary electrochromatography. | He, LX., et al. 2020. Electrophoresis. 41: 104-111. PMID: 31709552
- Trinuclear Lanthanide Coordination Clusters: Single-Molecule-Magnet Behavior and Catalytic Activity in the Friedel-Crafts Alkylation Reaction. | Sarkar, A., et al. 2019. Chempluschem. 84: 974-980. PMID: 31943977
- A simple organic probe for ratiometric fluorescent detection of Zn(II), Cd(II) and Hg(II) ions in aqueous solution via varying emission colours to distinguish one another. | Kumar, PS. and Elango, KP. 2020. Spectrochim Acta A Mol Biomol Spectrosc. 241: 118610. PMID: 32603882
- A New bis(rhodamine)-Based Colorimetric Chemosensor for Cu2. | Abebe, F., et al. 2020. Inorg Chem Commun. 120: PMID: 32863739
- New Dinuclear Macrocyclic Copper(II) Complexes as Potentially Fluorescent and Magnetic Materials. | Barwiolek, M., et al. 2023. Int J Mol Sci. 24: PMID: 36769351
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-tert-Butyl-2,6-diformylphenol, 1 g | sc-254725 | 1 g | $100.00 |
