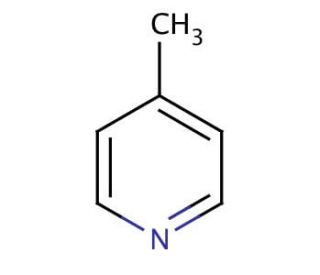
Molecular structure of 4-Picoline, CAS Number: 108-89-4
4-Picoline (CAS 108-89-4)
Alternate Names:
4-Methylpyridine
Application:
4-Picoline is a neutral N-donor ligand
CAS Number:
108-89-4
Molecular Weight:
93.13
Molecular Formula:
C6H7N
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Picoline is a chemical compound that functions as a precursor in the synthesis of agrochemicals and other organic compounds. It acts as a building block in the production of various heterocyclic compounds, including pyridine derivatives. 4-Picoline interacts with other reagents to undergo various chemical reactions, such as alkylation, acylation, and oxidation, to form complex organic molecules. Its mechanism of action involves participating in substitution reactions and serving as a starting material for the synthesis of diverse chemical compounds. 4-Picoline plays a role in the development of new materials and compounds with potential applications in various industries.
4-Picoline (CAS 108-89-4) References
- Laboratory evolution of toluene dioxygenase to accept 4-picoline as a substrate. | Sakamoto, T., et al. 2001. Appl Environ Microbiol. 67: 3882-7. PMID: 11525981
- Structural, Spectroscopic, and Magnetochemical Characterization of the Trinuclear Vanadium(III) Carboxylates [V(3)O(O(2)CR)(6)L(3)](ClO(4)) (R = Various Groups; L = Pyridine, 4-Picoline, 3,5-Lutidine). | Castro, SL., et al. 1996. Inorg Chem. 35: 4462-4468. PMID: 11666666
- The synthesis of 4-deazaformycin A. | Kourafalos, VN., et al. 2003. J Org Chem. 68: 6466-9. PMID: 12895092
- Enhanced expression, purification, and characterization of a novel class alpha glutathione S-transferase isozyme appearing in rabbit hepatic cytosol following treatment with 4-picoline. | Primiano, T. and Novak, RF. 1992. Toxicol Appl Pharmacol. 112: 291-9. PMID: 1539165
- Adsorption of 4-picoline and piperidine to the hydrated SiO2 surface: probing the surface acidity with vibrational sum frequency generation spectroscopy. | Liu, D., et al. 2005. Environ Sci Technol. 39: 2025-32. PMID: 15871233
- Transformation of 3- and 4-Picoline under Sulfate-Reducing Conditions. | Kaiser, JP., et al. 1993. Appl Environ Microbiol. 59: 701-5. PMID: 16348885
- Multinuclear NMR and theoretical investigation on interactions between diperoxovanadate complex and 4-picoline-like ligands. | Zhang, J., et al. 2010. Spectrochim Acta A Mol Biomol Spectrosc. 75: 83-7. PMID: 19900838
- Determination of pyridine, 2-picoline, 4-picoline and quinoline from mainstream cigarette smoke by solid-phase extraction liquid chromatography/electrospray ionization tandem mass spectrometry. | Saha, S., et al. 2010. J Chromatogr A. 1217: 307-11. PMID: 20004901
- 4,4',4'-trimethyl-2,2':6',2'-terpyridine by oxidative coupling of 4-picoline. | Robo, MT., et al. 2014. J Org Chem. 79: 10624-8. PMID: 25343728
- Synthesis of 4-benzylpyridines via Pd-catalyzed CH3-arylation of 4-picoline. | Wu, J., et al. 2017. Org Biomol Chem. 15: 7509-7512. PMID: 28862279
- Ruthenium-Catalyzed Oxidative Amidation of Alkynes to Amides. | Álvarez-Pérez, A., et al. 2019. Org Lett. 21: 5346-5350. PMID: 31247753
- Adsorptive separation of picoline isomers by adaptive calix[3]acridan crystals. | Zhou, HY. and Chen, CF. 2022. Chem Commun (Camb). 58: 4356-4359. PMID: 35293908
- C-Sulfonylation of 4-Alkylpyridines: Formal Picolyl C-H Activation via Alkylidene Dihydropyridine Intermediates. | Tun, SL., et al. 2023. J Org Chem. 88: 3998-4002. PMID: 36848377
Inhibitor of:
NOS3.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Picoline, 25 ml | sc-238951 | 25 ml | $28.00 |
