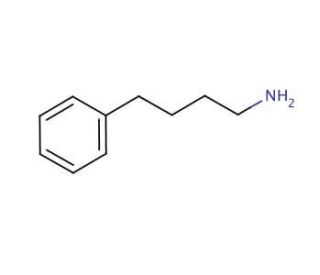
4-Phenylbutylamine (CAS 13214-66-9)
QUICK LINKS
4-Phenylbutylamine (4-PBA) is an organic compound extensively utilized in scientific research. Its versatile nature allows for various applications, including drug development, organic synthesis, and biochemistry. Belonging to the alkylamine family, 4-Phenylbutylamine is present in numerous natural products, such as the alkaloid ephedrine. With its capacity to interact with diverse molecules and relatively low toxicity, 4-Phenylbutylamine offers a broad spectrum of possibilities. It finds application in drug development, serves as a reagent in organic synthesis, and acts as a biochemical probe for investigating enzyme kinetics and protein-ligand interactions. Moreover, it serves as a valuable model compound for studying protein structure and function, as well as a substrate for exploring enzyme kinetics.
4-Phenylbutylamine (CAS 13214-66-9) References
- Cubic phases for studies of drug partition into lipid bilayers. | Engström, S., et al. 1999. Eur J Pharm Sci. 8: 243-54. PMID: 10425374
- Dimethyl ether chemical ionization of arylalkylamines. | Ramos, LE., et al. 2000. Rapid Commun Mass Spectrom. 14: 408-16. PMID: 10717649
- Structure-activity relations in the oxidation of phenethylamine analogues by recombinant human liver monoamine oxidase A. | Nandigama, RK. and Edmondson, DE. 2000. Biochemistry. 39: 15258-65. PMID: 11106506
- Solid phase synthesis of 4-hydroxycinnamic acid and its derivatives for potential use in combinatorial chemistry: a novel route for the synthesis of 4-hydroxycinnamoyl coenzyme A and NMDA receptor antagonists. | McIntyre, BG., et al. 2001. Comb Chem High Throughput Screen. 4: 111-4. PMID: 11281828
- [Application of solid-phase microextraction technique to the detection of amphetamines in urine by GC]. | Liu, W. and Shen, M. 1999. Fa Yi Xue Za Zhi. 15: 89-90, 127, 125. PMID: 12536407
- Inhibition of synaptosomal accumulation of l-norepinephrine II: N-aryloxyalkylphentermines, quaternary d-amphetamines, and 3-aryloxypropylamines. | Schaeffer, JC., et al. 1976. J Pharm Sci. 65: 122-6. PMID: 1255416
- The effect of amphiphilic phenylalkyl derivatives on platelet energy metabolism. Stimulation of glycolysis through activation of membrane ATPase. | Patscheke, H. and Brossmer, R. 1978. Arzneimittelforschung. 28: 1546-50. PMID: 157741
- Solvent-free derivatization of pristine multi-walled carbon nanotubes with amines. | Basiuk, EV., et al. 2005. J Nanosci Nanotechnol. 5: 984-90. PMID: 16060165
- Hybrid host-guest complexes: directing the supramolecular structure through secondary host-guest interactions. | Streb, C., et al. 2008. Chemistry. 14: 8861-8868. PMID: 18780383
- Degradation of human epidermal keratin by cod trypsin and extracts of fish intestines. | Bjelland, S., et al. 1989. Arch Dermatol Res. 280: 469-73. PMID: 2465741
- Synthesis, characterization, and application of a novel multifunctional stationary phase for hydrophilic interaction/reversed phase mixed-mode chromatography. | Aral, H., et al. 2017. Talanta. 174: 703-714. PMID: 28738646
- Molecular Disorder Induces an Unusual Phase Transition in a Potential 2D Chiral Ferroelectric Perovskite. | Sui Li, L., et al. 2021. Chemistry. 27: 9054-9059. PMID: 33847021
- Enhancement of alpha-chymotrypsin-catalyzed hydrolysis of specific p-nitroanilide substrates by 4-phenylbutylamine derivative of hen egg-white lysozyme. | Shechter, Y. and Gertler, A. 1978. Biochim Biophys Acta. 527: 42-55. PMID: 718965
- Partitioning of ionizing molecules between aqueous buffers and phospholipid vesicles. | Austin, RP., et al. 1995. J Pharm Sci. 84: 1180-3. PMID: 8801331
- Evaluation of affinity and pseudo-affinity adsorption processes for penicillin acylase purification. | Fonseca, LP. and Cabral, JM. 1996. Bioseparation. 6: 293-302. PMID: 9210349
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Phenylbutylamine, 5 g | sc-226776 | 5 g | $36.00 |
