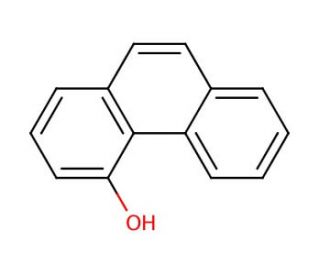
Molecular structure of 4-Phenanthrol, CAS Number: 7651-86-7
4-Phenanthrol (CAS 7651-86-7)
Alternate Names:
4-Phenanthrenol; 4-Hydroxyphenanthrene
Application:
4-Phenanthrol is a Phenanthrene metabolite
CAS Number:
7651-86-7
Molecular Weight:
194.23
Molecular Formula:
C14H10O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Phenanthrenol is a phenanthrene derivative found in many plants and fungi. It has antimicrobial, anti-inflammatory, antifungal, antioxidant, and antineoplastic activities. It inhibits the growth of bacteria and fungi by disrupting their cell walls and membranes. It is also interferes with the metabolism of bacteria and fungi, resulting in the inhibition of their growth. 4-Phenanthrenol has been employed in diverse scientific research, including studying antibiotic mechanisms, developing new antifungal agents, examining the biochemical and physiological effects of various compounds, and investigating enzyme structure and function.
4-Phenanthrol (CAS 7651-86-7) References
- Protein engineering of cytochrome p450(cam) (CYP101) for the oxidation of polycyclic aromatic hydrocarbons. | Harford-Cross, CF., et al. 2000. Protein Eng. 13: 121-8. PMID: 10708651
- Protein engineering of Bacillus megaterium CYP102. The oxidation of polycyclic aromatic hydrocarbons. | Carmichael, AB. and Wong, LL. 2001. Eur J Biochem. 268: 3117-25. PMID: 11358532
- Biodegradation of chloronaphthalenes and polycyclic aromatic hydrocarbons by the white-rot fungus Phlebia lindtneri. | Mori, T., et al. 2003. Appl Microbiol Biotechnol. 61: 380-3. PMID: 12743769
- The preparation of some metallized phenanthrene and naphthalene derivatives. | FOYE, WO., et al. 1952. J Am Pharm Assoc Am Pharm Assoc. 41: 312-5. PMID: 14946037
- Metabolite production in degradation of pyrene alone or in a mixture with another polycyclic aromatic hydrocarbon by Mycobacterium sp. | Zhong, Y., et al. 2006. Environ Toxicol Chem. 25: 2853-9. PMID: 17089707
- Metabolism of phenanthrene by Phanerochaete chrysosporium. | Sutherland, JB., et al. 1991. Appl Environ Microbiol. 57: 3310-6. PMID: 1781688
- Genome-to-function characterization of novel fungal P450 monooxygenases oxidizing polycyclic aromatic hydrocarbons (PAHs). | Syed, K., et al. 2010. Biochem Biophys Res Commun. 399: 492-7. PMID: 20674550
- Production of metabolites in the biodegradation of phenanthrene, fluoranthene and pyrene by the mixed culture of Mycobacterium sp. and Sphingomonas sp. | Zhong, Y., et al. 2011. Bioresour Technol. 102: 2965-72. PMID: 21036605
- Regiospecific oxidation of polycyclic aromatic phenols to quinones by hypervalent iodine reagents. | Wu, A., et al. 2010. Tetrahedron. 66: 2111-2118. PMID: 24014894
- Biodegradation of pyrene by pseudomonas sp. JPN2 and its initial degrading mechanism study by combining the catabolic nahAc gene and structure-based analyses. | Jin, J., et al. 2016. Chemosphere. 164: 379-386. PMID: 27596825
- Species-specific metabolism of naphthalene and phenanthrene in 3 species of marine teleosts exposed to Deepwater Horizon crude oil. | Pulster, EL., et al. 2017. Environ Toxicol Chem. 36: 3168-3176. PMID: 28636160
- Evaluation of fireground exposures using urinary PAH metabolites. | Hoppe-Jones, C., et al. 2021. J Expo Sci Environ Epidemiol. 31: 913-922. PMID: 33654270
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Phenanthrol, 5 mg | sc-391694 | 5 mg | $338.00 |
