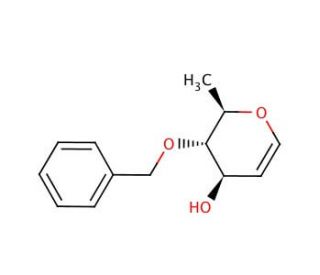
4-O-Benzyl-D-glucal (CAS 58871-11-7)
QUICK LINKS
4-O-Benzyl-D-glucal is a synthetic derivative of D-glucal, where the hydroxyl group at the fourth position is protected by a benzyl group. This compound is extensively used in carbohydrate chemistry and glycosylation reactions due to its ability to act as a glycosyl donor. The benzyl group provides stability, facilitating selective deprotection and subsequent chemical modifications. Researchers utilize 4-O-Benzyl-D-glucal to study the mechanisms of glycosylation enzymes, such as glycosyltransferases, which are critical for the formation of glycosidic bonds in the biosynthesis of complex carbohydrates and glycoconjugates. In particular, 4-O-Benzyl-D-glucal is used to synthesize oligosaccharides and glycoconjugates, enabling detailed studies of carbohydrate-protein interactions, which are vital for understanding cell-cell communication, immune response, and pathogen recognition. Additionally, this compound is instrumental in exploring the stereochemistry of glycosylation reactions, allowing researchers to investigate how different protecting groups influence the formation of glycosidic linkages. The benzyl group can be selectively removed under mild conditions, providing a versatile tool for the stepwise assembly of complex carbohydrates. Overall, 4-O-Benzyl-D-glucal is a valuable reagent in advancing the field of glycobiology, offering insights into the synthesis and function of glycan structures in biological systems, and aiding in the development of novel carbohydrate-based materials and probes.
4-O-Benzyl-D-glucal (CAS 58871-11-7) References
- Structural requirements of synthetic muropeptides to synergize with lipopolysaccharide in cytokine induction. | Traub, S., et al. 2004. J Biol Chem. 279: 8694-700. PMID: 14668350
- Total synthesis of N-acetylglucosamine-1,6-anhydro-N-acetylmuramylpentapeptide and evaluation of its turnover by AmpD from Escherichia coli. | Hesek, D., et al. 2009. J Am Chem Soc. 131: 5187-93. PMID: 19309146
- Versatile behavior of o-stannylated-d-glucal towards halogens. | Czernecki, Stanislas, et al. 1992. Tetrahedron letters. 33.2: 221-224.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-O-Benzyl-D-glucal, 250 mg | sc-256817 | 250 mg | $153.00 |
