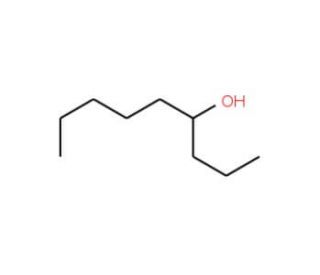
Molecular structure of 4-Nonanol, CAS Number: 5932-79-6
4-Nonanol (CAS 5932-79-6)
Alternate Names:
Amylpropylcarbinol; 4-Nonyl Alcohol; Pentylpropylcarbinol; Propylamylcarbinol; Propylpentylcarbinol
CAS Number:
5932-79-6
Molecular Weight:
144.25
Molecular Formula:
C9H20O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Nonanol, or nonan-4-ol, is an aliphatic alcohol (C9H20O) that is colorless and possesses a mild, sweet odor. It serves as a versatile solvent in various industrial applications and is notably utilized in liposomal drug delivery systems. This compound exhibits anti-inflammatory properties by diminishing the production of pro-inflammatory cytokines. Additionally, it possesses antioxidant capabilities by reducing the generation of reactive oxygen species.
4-Nonanol (CAS 5932-79-6) References
- Volatile compounds released by enzymatic hydrolysis of glycoconjugates of leaves and grape berries from Vitis vinifera Muscat of Alexandria and Shiraz cultivars. | Wirth, J., et al. 2001. J Agric Food Chem. 49: 2917-23. PMID: 11409987
- Changes in physicochemical characteristics and volatile constituents of strawberry (Cv. Cigaline) during maturation. | Ménager, I., et al. 2004. J Agric Food Chem. 52: 1248-54. PMID: 14995129
- The metabolism of nonane, a JP-8 jet fuel component, by human liver microsomes, P450 isoforms and alcohol dehydrogenase and inhibition of human P450 isoforms by JP-8. | Edwards, JE., et al. 2005. Chem Biol Interact. 151: 203-11. PMID: 15733541
- Changes in volatiles and glycosides during fruit maturation of two contrasted tomato (Solanum lycopersicum) lines. | Birtić, S., et al. 2009. J Agric Food Chem. 57: 591-8. PMID: 19154163
- Comparison of extraction methods for volatile compounds of Muscat grape juice. | Sánchez-Palomo, E., et al. 2009. Talanta. 79: 871-6. PMID: 19576458
- Changes in free and bound fractions of aroma compounds of four Vitis vinifera cultivars at the last ripening stages. | Vilanova, M., et al. 2012. Phytochemistry. 74: 196-205. PMID: 22071134
- Chemical composition and antimicrobial activity of the inflorescence essential oil of Capillipedium parviflorum (R. Br.) Stapf. from India. | Verma, RS., et al. 2012. Nat Prod Res. 26: 1257-60. PMID: 22077331
- Identification of volatile compounds associated with the aroma of white strawberries (Fragaria chiloensis). | Prat, L., et al. 2014. J Sci Food Agric. 94: 752-9. PMID: 24115051
- Aroma enhancement of cherry juice and wine using exogenous glycosidases from mould, yeast and lactic acid bacteria. | Wilkowska, A. and Pogorzelski, E. 2017. Food Chem. 237: 282-289. PMID: 28763997
- Effect of seasonal climate fluctuations on the evolution of glycoconjugates during the ripening period of grapevine cv. Muscat à petits grains blancs berries. | Crespo, J., et al. 2018. J Sci Food Agric. 98: 1803-1812. PMID: 28873234
- New PLS analysis approach to wine volatile compounds characterization by near infrared spectroscopy (NIR). | Genisheva, Z., et al. 2018. Food Chem. 246: 172-178. PMID: 29291836
- Assessment fertigation effects on chemical composition of Vitis vinifera L. cv. Albariño. | Vilanova, M., et al. 2019. Food Chem. 278: 636-643. PMID: 30583423
- Interface mechanisms of the catalytic ozonation of humic acids over siliceous ferrihydrite: Morphology, stability, and the catalytic process. | Chen, L., et al. 2022. Environ Res. 203: 111870. PMID: 34390719
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Nonanol, 5 g | sc-277586 | 5 g | $208.00 |
