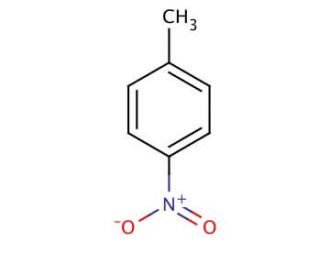
4-Nitrotoluene (CAS 99-99-0)
QUICK LINKS
4-Nitrotoluene is a para-substituted toluene for proteomics research 4-Nitrotoluene stands as an aromatic compound with extensive utility in crafting a diverse range of organic substances. This colorless solid emits a potent, piquant scent and exhibits high flammability. The production of 4-Nitrotoluene involves the nitration of toluene using nitric acid. This compound assumes a pivotal role as an intermediary in the creation of dyes and explosives, while also finding application in the fabrication of polyurethanes, polyesters, and polyamides. Beyond its industrial applications, 4-Nitrotoluene has found its way into various scientific research undertakings. Its involvement in dye synthesis, as well as its contribution to explosive creation, showcases its multifaceted potential. Notably, the compound plays a part in generating polyurethanes, polyesters, and polyamides. Moreover, it plays a role in the synthesis of nitroaromatics, components in the construction of explosives, propellants, and munitions. The precise mechanism by which 4-Nitrotoluene operates remains somewhat enigmatic. However, prevailing theories attribute its reactivity to the nitro group within the molecule. Functioning as a potent electron-withdrawing entity, this nitro group engages in reactions with other molecules, heightening their reactivity. This inherent reactivity positions 4-Nitrotoluene as useful for synthesizing an array of organic compounds.
4-Nitrotoluene (CAS 99-99-0) References
- A two-generation reproductive toxicity study of 4-nitrotoluene in rats. | Aso, S., et al. 2005. J Toxicol Sci. 30 Spec No.: 117-134. PMID: 16641538
- Stepwise oxygenations of toluene and 4-nitrotoluene by a fungal peroxygenase. | Kinne, M., et al. 2010. Biochem Biophys Res Commun. 397: 18-21. PMID: 20470751
- Isolation and characterization of a 4-nitrotoluene-oxidizing enzyme from activated sludge by a metagenomic approach. | Kimura, N., et al. 2010. Microbes Environ. 25: 133-9. PMID: 21576864
- Evolution of a new bacterial pathway for 4-nitrotoluene degradation. | Ju, KS. and Parales, RE. 2011. Mol Microbiol. 82: 355-64. PMID: 21895789
- Biodegradation of 4-nitrotoluene with biosurfactant production by Rhodococcus pyridinivorans NT2: metabolic pathway, cell surface properties and toxicological characterization. | Kundu, D., et al. 2013. Biodegradation. 24: 775-93. PMID: 23389716
- Resolving Discrepancy between Theory and Experiment in 4-Nitrotoluene Oxidation. | Klajman, K., et al. 2017. J Phys Chem A. 121: 6638-6645. PMID: 28806085
- Dissociation dynamics of 3- and 4-nitrotoluene radical cations: Coherently driven C-NO2 bond homolysis. | Ampadu Boateng, D., et al. 2018. J Chem Phys. 148: 134305. PMID: 29626885
- Pyrenyl-Functionalized Polysiloxane Based on Synergistic Effect for Highly Selective and Highly Sensitive Detection of 4-Nitrotoluene. | Gou, Z., et al. 2019. ACS Appl Mater Interfaces. 11: 30218-30227. PMID: 31356040
- Spherical silver oxide nanoparticles for fabrication of electrochemical sensor for efficient 4-Nitrotoluene detection and assessment of their antimicrobial activity. | Chakraborty, U., et al. 2022. Sci Total Environ. 808: 152179. PMID: 34875317
- Fabrication of Carbon Nanofiber Incorporated with CuWO4 for Sensitive Electrochemical Detection of 4-Nitrotoluene in Water Samples. | Meenakshi, GA., et al. 2023. Sensors (Basel). 23: PMID: 37420832
- A novel pathway for the catabolism of 4-nitrotoluene by Pseudomonas. | Rhys-Williams, W., et al. 1993. J Gen Microbiol. 139: 1967-72. PMID: 8245826
- Biodegradation of 4-nitrotoluene by Pseudomonas sp. strain 4NT. | Haigler, BE. and Spain, JC. 1993. Appl Environ Microbiol. 59: 2239-43. PMID: 8357257
- In vivo construction of a hybrid pathway for metabolism of 4-nitrotoluene in Pseudomonas fluorescens. | Michán, C., et al. 1997. J Bacteriol. 179: 3036-8. PMID: 9139924
- A new 4-nitrotoluene degradation pathway in a Mycobacterium strain. | Spiess, T., et al. 1998. Appl Environ Microbiol. 64: 446-52. PMID: 9464378
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Nitrotoluene, 100 g | sc-238935 | 100 g | $15.00 |
