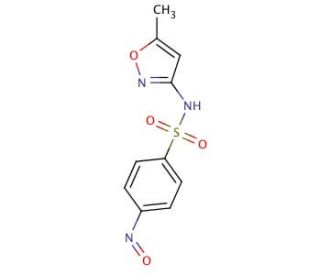
Molecular structure of 4-Nitroso Sulfamethoxazole, CAS Number: 131549-85-4�
4-Nitroso Sulfamethoxazole (CAS 131549-85-4)
Alternate Names:
N-(5-methyl-1,2-oxazol-3-yl)-4-nitrosobenzenesulfonamide
Application:
4-Nitroso Sulfamethoxazole is a Sulfamethoxazole metabolite
CAS Number:
131549-85-4
Purity:
≥90%
Molecular Weight:
267.26
Molecular Formula:
C10H9N3O4S
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Nitroso Sulfamethoxazole is a Sulfamethoxazole metabolite. 4-Nitroso Sulfamethoxazole exhibits inhibitory properties against dihydropteroate synthase, a enzyme in the folate biosynthesis pathway. This compound serves as useful in exploring the significance of folate metabolism in diverse biological processes. Its application includes investigating the impacts of folate deficiency on DNA methylation and gene expression. Furthermore, it has been utilized in studying the involvement of folate metabolism in cancer cells.
4-Nitroso Sulfamethoxazole (CAS 131549-85-4) References
- Direct photolysis of human metabolites of the antibiotic sulfamethoxazole: evidence for abiotic back-transformation. | Bonvin, F., et al. 2013. Environ Sci Technol. 47: 6746-55. PMID: 23186099
- Systematic suspect screening and identification of sulfonamide antibiotic transformation products in the aquatic environment. | Majewsky, M., et al. 2015. Anal Bioanal Chem. 407: 5707-17. PMID: 26070765
- Phototransformation of sulfamethoxazole under simulated sunlight: Transformation products and their antibacterial activity toward Vibrio fischeri. | Gmurek, M., et al. 2015. Sci Total Environ. 538: 58-63. PMID: 26298248
- Biodegradation of sulfamethoxazole photo-transformation products in a water/sediment test. | Su, T., et al. 2016. Chemosphere. 148: 518-25. PMID: 26845465
- 4-nitroso-sulfamethoxazole generation in soil under denitrifying conditions: Field observations versus laboratory results. | Brienza, M., et al. 2017. J Hazard Mater. 334: 185-192. PMID: 28412628
- Improvement of sulfamethoxazole (SMX) elimination and inhibition of formations of hydroxylamine-SMX and N4-acetyl-SMX by membrane bioreactor systems. | Tang, MH., et al. 2018. Biodegradation. 29: 245-258. PMID: 29546497
- Non-activated peroxymonosulfate oxidation of sulfonamide antibiotics in water: Kinetics, mechanisms, and implications for water treatment. | Ji, Y., et al. 2018. Water Res. 147: 82-90. PMID: 30300784
- Fate of 14C-acetyl sulfamethoxazole during the activated sludge process. | Geng, C., et al. 2019. Environ Sci Pollut Res Int. 26: 9832-9841. PMID: 30734913
- Whole genome sequencing identifies genetic variants associated with co-trimoxazole hypersensitivity in Asians. | Wang, CW., et al. 2021. J Allergy Clin Immunol. 147: 1402-1412. PMID: 32791162
- Direct oxidation of antibiotic trimethoprim by unactivated peroxymonosulfate via a nonradical transformation mechanism. | Yang, X., et al. 2021. Chemosphere. 263: 128194. PMID: 33297159
- Accurate Quantification of Sulfonamide Metabolites in Goat Meat: A New Strategy for Minimizing Interaction between Sheep Serum Albumin and Sulfonamide Metabolites. | Jia, W., et al. 2021. J Agric Food Chem. 69: 6556-6568. PMID: 34080416
- Insights on the role of periphytic biofilm in synergism with Iris pseudacorus for removing mixture of pharmaceutical contaminants from wastewater. | Yadav, N., et al. 2021. J Hazard Mater. 418: 126349. PMID: 34118536
- Micropollutant removal and disinfection byproduct control by sequential peroxymonosulfate-UV treatment in water: A case study with sulfamethoxazole. | Zhang, TY., et al. 2022. J Environ Sci (China). 117: 141-150. PMID: 35725066
- Isotopically labeled ozone: A new approach to elucidate the formation of ozonation products. | Sierra-Olea, M., et al. 2023. Water Res. 233: 119740. PMID: 36822109
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Nitroso Sulfamethoxazole, 2.5 mg | sc-210132 | 2.5 mg | $406.00 |
