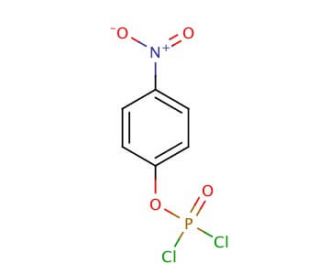
4-Nitrophenyl phosphorodichloridate (CAS 777-52-6)
QUICK LINKS
4-Nitrophenyl phosphorodichloridate is a powerful phosphorylating agent used for nucleosides. 4-Nitrophenyl phosphorodichloridate, referred to as 4-NPP or PNP, is a versatile organophosphate compound extensively utilized in scientific research. As a colorless, odorless, and water-soluble solid, it finds wide-ranging applications in laboratory experiments and various scientific endeavors. Its primary role lies in inhibiting enzymes, making it useful in research. By acting as an inhibitor, 4-Nitrophenyl phosphorodichloridate effectively hampers the activity of enzymes, including acetylcholinesterase and thymidylate synthase. Furthermore, it can impede other enzyme-catalyzed reactions such as DNA polymerase, while also serving as a substrate for diverse enzymes. Consequently, this compound has proved instrumental in investigating enzyme kinetics, unraveling protein-protein interactions, and exploring other processes. The mechanism of action for 4-Nitrophenyl phosphorodichloridate involves binding to the active site of enzymes, thereby obstructing the entry of substrates. By doing so, it impedes the catalyzing function of enzymes. This inhibition of enzyme activity allows for in-depth exploration of enzyme function, regulation, and their interplay with other proteins. Thus, 4-Nitrophenyl phosphorodichloridate provides a valuable avenue for studying various aspects of enzymology and elucidating intricate biochemical processes.
4-Nitrophenyl phosphorodichloridate (CAS 777-52-6) References
- A synthetic study on cyclic phosphate derivatives of seconucleosides as potential antiviral agents (I). Synthesis of 3',5'-cyclic phosphates of 2'-substituted secouridines and secoribavirins. | Hong, KA., et al. 1991. Arch Pharm Res. 14: 30-4. PMID: 10319117
- A comparison of flexible and constrained haptens in eliciting antibody catalysts for paraoxon hydrolysis. | Spivak, DA., et al. 1999. Bioorg Med Chem. 7: 1145-50. PMID: 10428386
- Synthesis and Kinetic Evaluation of Inhibitors of the Phosphatidylinositol-Specific Phospholipase C from Bacillus cereus. | Martin, SF. and Wagman, AS. 1996. J Org Chem. 61: 8016-8023. PMID: 11667784
- Synthesis and mass spectrometry of crufomate metabolites and related compounds. | Feil, VJ., et al. 1976. Biomed Mass Spectrom. 3: 316-28. PMID: 137004
- Synthesis and characterization of a novel liver-targeted prodrug of cytosine-1-beta-D-arabinofuranoside monophosphate for the treatment of hepatocellular carcinoma. | Boyer, SH., et al. 2006. J Med Chem. 49: 7711-20. PMID: 17181153
- A chromogenic substrate for phosphatidylinositol-specific phospholipase C: 4-nitrophenyl myo-inositol-1-phosphate. | Shashidhar, MS., et al. 1991. Chem Phys Lipids. 60: 101-10. PMID: 1814635
- A polyclonal antibody preparation with Michaelian catalytic properties. | Gallacher, G., et al. 1991. Biochem J. 279 (Pt 3): 871-81. PMID: 1953683
- Synthesis, spectral characterization and biological evaluation of phosphorylated derivatives of galanthamine. | Koteswara Rao, V., et al. 2010. Eur J Med Chem. 45: 203-9. PMID: 19853328
- Amino acid esters substituted phosphorylated emtricitabine and didanosine derivatives as antiviral and anticancer agents. | Sekhar, KC., et al. 2014. Appl Biochem Biotechnol. 173: 1303-18. PMID: 24789416
- Molecular self-assembly of amphiphilic cyclic phosphoryl gemcitabine with different N-fatty acyl tails and enhanced anticancer effects of the self-assembled nanostructures. | Jin, Y., et al. 2015. Colloids Surf B Biointerfaces. 133: 356-61. PMID: 26143151
- In silico docking studies and synthesis of new phosphoramidate derivatives of 6-fluoro-3-(piperidin-4-yl)benzo[d]isoxazole as potential antimicrobial agents. | Sivala, MR., et al. 2020. J Recept Signal Transduct Res. 40: 486-492. PMID: 32312118
- Random and site-specific immobilization of catalytic antibodies. | Spitznagel, TM., et al. 1993. Enzyme Microb Technol. 15: 916-21. PMID: 7764251
- Improved synthesis of myo-inositol 1-(4-nitrophenyl hydrogen phosphate), a chromogenic substrate for phosphatidylinositol-specific phospholipase C. | Rukavishnikov, AV., et al. 1997. Chem Phys Lipids. 89: 153-7. PMID: 9369010
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Nitrophenyl phosphorodichloridate, 5 g | sc-256812 | 5 g | $35.00 |
