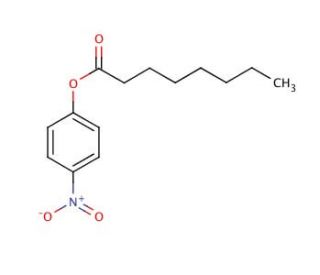
4-Nitrophenyl octanoate (CAS 1956-10-1)
QUICK LINKS
4-Nitrophenyl octanoate (4-NPCL) is a synthetic compound formed by the esterification of 4-nitrophenol and caprylic acid. The utilization of 4-Nitrophenyl octanoate spans across diverse scientific research applications. Notably, this versatile intermediate finds application as a reagent for detecting enzymes such as lipases, esterases, and phosphatases. Furthermore, 4-Nitrophenyl octanoate plays a vital role in the synthesis of various compounds. The precise mechanism of action of 4-Nitrophenyl octanoate remains under investigation. However, current understanding suggests that the esterification reaction between 4-nitrophenol and caprylic acid is catalyzed by an acid catalyst, resulting in the release of nitric oxide, considered the active species. Furthermore, it is hypothesized that nitric oxide interacts with the substrate, leading to the formation of 4-Nitrophenyl octanoate.
4-Nitrophenyl octanoate (CAS 1956-10-1) References
- [Cloning, expression and characterization of a novel lipase gene lipB from Aspergillus niger F044]. | Yang, J., et al. 2009. Wei Sheng Wu Xue Bao. 49: 1095-101. PMID: 19835173
- Molecular and biochemical diversity of Oenococcus oeni strains isolated during spontaneous malolactic fermentation of Malvasia Nera wine. | Cappello, MS., et al. 2010. Syst Appl Microbiol. 33: 461-7. PMID: 21095085
- The σB-dependent yabJ-spoVG operon is involved in the regulation of extracellular nuclease, lipase, and protease expression in Staphylococcus aureus. | Schulthess, B., et al. 2011. J Bacteriol. 193: 4954-62. PMID: 21725011
- Isolation and characterization of an enzyme from the Greenshell™ mussel Perna canaliculus that hydrolyses pectenotoxins and esters of okadaic acid. | MacKenzie, LA., et al. 2012. Toxicon. 60: 406-19. PMID: 22613166
- In vitro investigation of the potential health benefits of wild Mediterranean dietary plants as anti-obesity agents with α-amylase and pancreatic lipase inhibitory activities. | Marrelli, M., et al. 2014. J Sci Food Agric. 94: 2217-24. PMID: 24535986
- Characterisation of a New Family of Carboxyl Esterases with an OsmC Domain. | Jensen, MV., et al. 2016. PLoS One. 11: e0166128. PMID: 27851780
- Biological Activities of Camelina and Sophia Seeds Phenolics: Inhibition of LDL Oxidation, DNA Damage, and Pancreatic Lipase and α-Glucosidase Activities. | Rahman, MJ., et al. 2018. J Food Sci. 83: 237-245. PMID: 29278656
- Non-proteinaceous hydrolase comprised of a phenylalanine metallo-supramolecular amyloid-like structure. | Makam, P., et al. 2019. Nat Catal. 2: 977-985. PMID: 31742246
- Discovery and characterization of a stable lipase with preference toward long-chain fatty acids. | Chen, M., et al. 2020. Biotechnol Lett. 42: 171-180. PMID: 31745842
- A rapid millifluidic synthesis of tunable polymer-protein nanoparticles. | Seaberg, J., et al. 2020. Eur J Pharm Biopharm. 154: 127-135. PMID: 32659325
- Amphipathic Janus Membrane with Hierarchical Multiscale Hyperporous Structure for Interfacial Catalysis. | Lin, Y., et al. 2020. Membranes (Basel). 10: PMID: 32717990
- Lipase inhibitory activity assay for fermented milk. | Gil-Rodríguez, AM. and Beresford, TP. 2020. MethodsX. 7: 100999. PMID: 32775223
- An ultrahigh-throughput screening platform based on flow cytometric droplet sorting for mining novel enzymes from metagenomic libraries. | Ma, F., et al. 2021. Environ Microbiol. 23: 996-1008. PMID: 32985743
- Characterisation of a Novel Acetyl Xylan Esterase (BaAXE) Screened from the Gut Microbiota of the Common Black Slug (Arion ater). | Madubuike, H. and Ferry, N. 2022. Molecules. 27: PMID: 35566348
- Plant biochemistry of xenobiotics. Purification and properties of a wheat esterase hydrolyzing the plasticizer chemical, bis(2-ethylhexyl)phthalate. | Krell, HW. and Sandermann, H. 1984. Eur J Biochem. 143: 57-62. PMID: 6468391
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Nitrophenyl octanoate, 1 g | sc-210130 | 1 g | $41.00 | |||
4-Nitrophenyl octanoate, 5 g | sc-210130A | 5 g | $84.00 |
