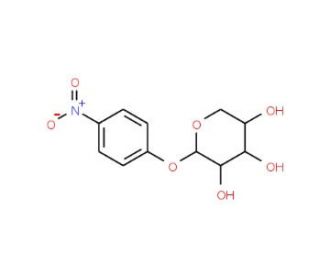
Molecular structure of 4-Nitrophenyl-β-D- xylopyranoside, CAS Number: 2001-96-9
4-Nitrophenyl-β-D- xylopyranoside (CAS 2001-96-9)
Alternate Names:
PNPX; p-Nitrophenyl-β-D-xylopyranoside; PNP-β-D-Xyl
Application:
4-Nitrophenyl-β-D- xylopyranoside is a chromogenic β-xylosidase substrate
CAS Number:
2001-96-9
Purity:
≥99%
Molecular Weight:
271.22
Molecular Formula:
C11H13NO7
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Nitrophenyl-β-D-xylopyranoside is a substrate biochemical to study the activity of enzymes such as β-xylosidase. In enzymatic assays, 4-Nitrophenyl-β-D-xylopyranoside is hydrolyzed by β-xylosidase to release 4-nitrophenol, which can be detected spectrophotometrically. This allows for the measurement of enzyme activity and the determination of kinetic parameters. The ability to monitor enzyme activity makes it useful in the studying carbohydrate metabolism and the development of enzyme inhibitors. 4-Nitrophenyl-β-D-xylopyranoside has good stability and reliability in biochemical assays.
4-Nitrophenyl-β-D- xylopyranoside (CAS 2001-96-9) References
- Lipase-catalysed preparation of acetates of 4-nitrophenyl beta-D-xylopyranoside and their use in kinetic studies of acetyl migration. | Mastihubová, M. and Biely, P. 2004. Carbohydr Res. 339: 1353-60. PMID: 15113674
- Enzyme-coupled assay of acetylxylan esterases on monoacetylated 4-nitrophenyl beta-D-xylopyranosides. | Biely, P., et al. 2004. Anal Biochem. 332: 109-15. PMID: 15301955
- Monitoring carbohydrate enzymatic reactions by quantitative in vitro microdialysis. | Modi, SJ. and LaCourse, WR. 2006. J Chromatogr A. 1118: 125-33. PMID: 16546198
- The vicinal hydroxyl group is prerequisite for metal activation of Clostridium thermocellum acetylxylan esterase. | Biely, P., et al. 2007. Biochim Biophys Acta. 1770: 565-70. PMID: 17261352
- Variation in relative substrate specificity of bifunctional beta-D-xylosidase/alpha-L-arabinofuranosidase by single-site mutations: roles of substrate distortion and recognition. | Jordan, DB. and Li, XL. 2007. Biochim Biophys Acta. 1774: 1192-8. PMID: 17689155
- Aminoalcohols as probes of the two-subsite active site of beta-D-xylosidase from Selenomonas ruminantium. | Jordan, DB., et al. 2009. Biochim Biophys Acta. 1794: 144-58. PMID: 18973836
- Action of xylan deacetylating enzymes on monoacetyl derivatives of 4-nitrophenyl glycosides of β-D-xylopyranose and α-L-arabinofuranose. | Biely, P., et al. 2011. J Biotechnol. 151: 137-42. PMID: 21029756
- Interaction of Listeria monocytogenes autolysin amidase with glycosaminoglycans promotes listerial adhesion to mouse hepatocytes. | Asano, K., et al. 2012. Biochimie. 94: 1291-9. PMID: 22386869
- Positional specifity of acetylxylan esterases on natural polysaccharide: an NMR study. | Uhliariková, I., et al. 2013. Biochim Biophys Acta. 1830: 3365-72. PMID: 23375723
- Rate-limiting steps of a stereochemistry retaining β-d-xylosidase from Geobacillus stearothermophilus acting on four substrates. | Jordan, DB. and Braker, JD. 2015. Arch Biochem Biophys. 583: 73-8. PMID: 26271441
- A novel thermostable GH5 β-xylosidase from Thermogemmatispora sp. T81. | Tomazini, A., et al. 2019. N Biotechnol. 53: 57-64. PMID: 31299302
- Functional screening of a Caatinga goat (Capra hircus) rumen metagenomic library reveals a novel GH3 β-xylosidase. | Souto, BM., et al. 2021. PLoS One. 16: e0245118. PMID: 33449963
- The beta-D-xylosidase of Trichoderma reesei is a multifunctional beta-D-xylan xylohydrolase. | Herrmann, MC., et al. 1997. Biochem J. 321 (Pt 2): 375-81. PMID: 9020869
Inhibitor of:
chondroitin sulfate.Substrate of:
Chromogenic, and Enzyme.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Nitrophenyl-β-D- xylopyranoside, 500 mg | sc-281430 | 500 mg | $268.00 | |||
4-Nitrophenyl-β-D- xylopyranoside, 1 g | sc-281430A | 1 g | $493.00 |
