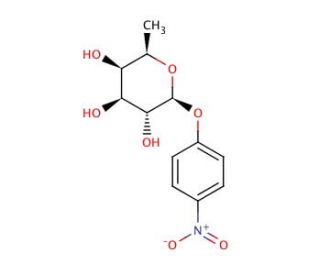
Molecular structure of 4-Nitrophenyl β-D-fucopyranoside, CAS Number: 1226-39-7
4-Nitrophenyl β-D-fucopyranoside (CAS 1226-39-7)
Alternate Names:
PNP-b-D-Fuc
Application:
4-Nitrophenyl β-D-fucopyranoside is a chromogenic substrate
CAS Number:
1226-39-7
Purity:
≥97%
Molecular Weight:
285.25
Molecular Formula:
C12H15NO7
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Nitrophenyl β-D-fucopyranoside is a potent enzyme protein glycosylation inhibitor. It exhibits the ability of inhibiting bacterial growth, both Gram-positive and Gram-negative species. The sequenced strain is able to grow on solar and alcohol substrates, while bacterial strain affinity depends on morphology. Remarkably, the amino acid sequence of 4-Nitrophenyl β-D-fucopyranoside closely resembles that of agarose and transferase, both pivotal enzymes within glycosylation processes. 4-Nitrophenyl β-D-fucopyranoside serves as a clone and cosmogenic marker in electron microscopy studies.
4-Nitrophenyl β-D-fucopyranoside (CAS 1226-39-7) References
- Directed evolution of a glycosynthase from Agrobacterium sp. increases its catalytic activity dramatically and expands its substrate repertoire. | Kim, YW., et al. 2004. J Biol Chem. 279: 42787-93. PMID: 15252054
- DeltaG(CH2) as solvent descriptor in polymer/polymer aqueous two-phase systems. | Madeira, PP., et al. 2008. J Chromatogr A. 1185: 85-92. PMID: 18258243
- Glycolytic cancer cells lacking 6-phosphogluconate dehydrogenase metabolize glucose to induce senescence. | Sukhatme, VP. and Chan, B. 2012. FEBS Lett. 586: 2389-95. PMID: 22677172
- Characterization of new β-galactosidase from acidophilic fungus, Teratosphaeria acidotherma AIU BGA-1. | Isobe, K., et al. 2013. J Biosci Bioeng. 116: 293-7. PMID: 23623895
- New alkalophilic β-galactosidase with high activity in alkaline pH region from Teratosphaeria acidotherma AIU BGA-1. | Yamada, M., et al. 2017. J Biosci Bioeng. 123: 15-19. PMID: 27514907
- Biochemical Characterization and Low-Resolution SAXS Molecular Envelope of GH1 β-Glycosidase from Saccharophagus degradans. | Brognaro, H., et al. 2016. Mol Biotechnol. 58: 777-788. PMID: 27670285
- Structural insights into β-glucosidase transglycosylation based on biochemical, structural and computational analysis of two GH1 enzymes from Trichoderma harzianum. | Florindo, RN., et al. 2018. N Biotechnol. 40: 218-227. PMID: 28888962
- Glycoside hydrolase DisH from Desulfovibrio vulgaris degrades the N-acetylgalactosamine component of diverse biofilms. | Zhu, L., et al. 2018. Environ Microbiol. 20: 2026-2037. PMID: 29411481
- Structural and biochemical characterization of a GH3 β-glucosidase from the probiotic bacteria Bifidobacterium adolescentis. | Florindo, RN., et al. 2018. Biochimie. 148: 107-115. PMID: 29555372
- A glucotolerant β-glucosidase from the fungus Talaromyces amestolkiae and its conversion into a glycosynthase for glycosylation of phenolic compounds. | Méndez-Líter, JA., et al. 2020. Microb Cell Fact. 19: 127. PMID: 32522206
- Accounting for the instantaneous disorder in the enzyme-substrate Michaelis complex to calculate the Gibbs free energy barrier of an enzyme reaction. | Romero-Téllez, S., et al. 2021. Phys Chem Chem Phys. 23: 13042-13054. PMID: 34100037
- Solute partitioning in polymer–salt ATPS: The Collander equation | Sara C. Silvério a b, Oscar Rodriguez a, José A. Teixeira b, Eugénia A. Macedo a. 2010. Fluid Phase Equilibria. 296: 173-177.
- β-Galactosidases from an Acidophilic Fungus, Teratosphaeria acidotherma AIU BGA-1 | Kimiyasu Isobe & Miwa Yamada. 2019. Fungi in Extreme Environments: Ecological Role and Biotechnological Significance. 419–440.
Substrate of:
Chromogenic.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Nitrophenyl β-D-fucopyranoside, 25 mg | sc-216968 | 25 mg | $42.00 | |||
4-Nitrophenyl β-D-fucopyranoside, 100 mg | sc-216968A | 100 mg | $75.00 |
