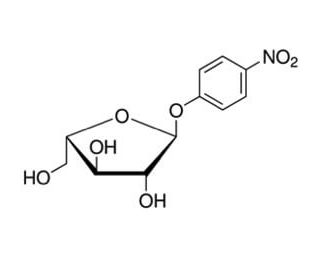
Molecular structure of 4-Nitrophenyl α-L-arabinofuranoside, CAS Number: 6892-58-6
4-Nitrophenyl α-L-arabinofuranoside (CAS 6892-58-6)
See product citations (1)
Alternate Names:
pNP-alpha-L-Arabinofuranoside
CAS Number:
6892-58-6
Purity:
≥98%
Molecular Weight:
271.22
Molecular Formula:
C11H13NO7
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Nitrophenyl α-L-arabinofuranoside is a synthetic compound often used in biochemistry and molecular biology as a chromogenic substrate in the assay of α-L-arabinofuranosidase activity. α-L-Arabinofuranosidases are enzymes that catalyze the hydrolysis of α-L-arabinofuranoside bonds, which are found in various natural compounds, including polysaccharides and glycoconjugates. These enzymes play a role in the breakdown and metabolism of complex carbohydrates, contributing to the degradation of plant biomass and the cycling of organic matter in the environment.
4-Nitrophenyl α-L-arabinofuranoside (CAS 6892-58-6) References
- Bifunctional family 3 glycoside hydrolases from barley with alpha -L-arabinofuranosidase and beta -D-xylosidase activity. Characterization, primary structures, and COOH-terminal processing. | Lee, RC., et al. 2003. J Biol Chem. 278: 5377-87. PMID: 12464603
- The acetates of p-nitrophenyl alpha-L-arabinofuranoside--regioselective preparation by action of lipases. | Mastihubová, M., et al. 2006. Bioorg Med Chem. 14: 1805-10. PMID: 16288881
- Variation in relative substrate specificity of bifunctional beta-D-xylosidase/alpha-L-arabinofuranosidase by single-site mutations: roles of substrate distortion and recognition. | Jordan, DB. and Li, XL. 2007. Biochim Biophys Acta. 1774: 1192-8. PMID: 17689155
- The construction and characterization of two xylan-degrading chimeric enzymes. | Fan, Z., et al. 2009. Biotechnol Bioeng. 102: 684-92. PMID: 18828173
- Thermodynamics of the hydrolysis reactions of 1-naphthyl acetate, 4-nitrophenyl acetate, and 4-nitrophenyl α-L-arabinofuranoside. | Decker, SR., et al. 2010. J Phys Chem B. 114: 16060-7. PMID: 20361764
- Novel bifunctional alpha-L-arabinofuranosidase/xylobiohydrolase (ABF3) from Penicillium purpurogenum. | Ravanal, MC., et al. 2010. Appl Environ Microbiol. 76: 5247-53. PMID: 20562284
- Action of xylan deacetylating enzymes on monoacetyl derivatives of 4-nitrophenyl glycosides of β-D-xylopyranose and α-L-arabinofuranose. | Biely, P., et al. 2011. J Biotechnol. 151: 137-42. PMID: 21029756
- Purification and characterization of arabinofuranosidase from the corn endophyte Acremonium zeae. | Bischoff, KM., et al. 2011. Biotechnol Lett. 33: 2013-8. PMID: 21671092
- Analysis of the arabinoxylan arabinofuranohydrolase gene family in barley does not support their involvement in the remodelling of endosperm cell walls during development. | Laidlaw, HK., et al. 2012. J Exp Bot. 63: 3031-45. PMID: 22378943
- Divalent metal activation of a GH43 β-xylosidase. | Lee, CC., et al. 2013. Enzyme Microb Technol. 52: 84-90. PMID: 23273276
- A novel bifunctional GH51 exo-α-l-arabinofuranosidase/endo-xylanase from Alicyclobacillus sp. A4 with significant biomass-degrading capacity. | Yang, W., et al. 2015. Biotechnol Biofuels. 8: 197. PMID: 26628911
- Characterization of a putative glycoside hydrolase family 43 arabinofuranosidase from Aspergillus niger and its potential use in beer production. | Li, X., et al. 2020. Food Chem. 305: 125382. PMID: 31525590
- Characterization and functional analysis of two novel thermotolerant α-L-arabinofuranosidases belonging to glycoside hydrolase family 51 from Thielavia terrestris and family 62 from Eupenicillium parvum. | Long, L., et al. 2020. Appl Microbiol Biotechnol. 104: 8719-8733. PMID: 32880690
- The beta-D-xylosidase of Trichoderma reesei is a multifunctional beta-D-xylan xylohydrolase. | Herrmann, MC., et al. 1997. Biochem J. 321 (Pt 2): 375-81. PMID: 9020869
- Purification and characterization of a novel thermostable alpha-L-arabinofuranosidase from a color-variant strain of Aureobasidium pullulans. | Saha, BC. and Bothast, RJ. 1998. Appl Environ Microbiol. 64: 216-20. PMID: 9435077
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Nitrophenyl α-L-arabinofuranoside, 10 mg | sc-220982 | 10 mg | $109.00 | |||
4-Nitrophenyl α-L-arabinofuranoside, 25 mg | sc-220982A | 25 mg | $203.00 | |||
4-Nitrophenyl α-L-arabinofuranoside, 50 mg | sc-220982B | 50 mg | $359.00 | |||
4-Nitrophenyl α-L-arabinofuranoside, 100 mg | sc-220982C | 100 mg | $515.00 | |||
4-Nitrophenyl α-L-arabinofuranoside, 250 mg | sc-220982D | 250 mg | $983.00 |
