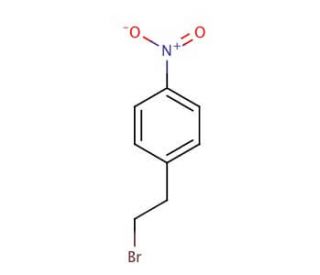
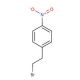
Molecular structure of 4-Nitrophenethyl bromide, CAS Number: 5339-26-4
4-Nitrophenethyl bromide (CAS 5339-26-4)
Alternate Names:
1-(2-Bromoethyl)-4-nitrobenzene; 2-(4-Nitrophenyl)ethyl bromide
Application:
4-Nitrophenethyl bromide is a GST T1-1 substrate
CAS Number:
5339-26-4
Purity:
≥98%
Molecular Weight:
230.06
Molecular Formula:
C8H8BrNO2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Nitrophenethyl bromide is an electrophile used as a substrate and activator for theta class gutathione transferase (GSTT-1).
4-Nitrophenethyl bromide (CAS 5339-26-4) References
- Molecular identification of cytosolic prostaglandin E2 synthase that is functionally coupled with cyclooxygenase-1 in immediate prostaglandin E2 biosynthesis. | Tanioka, T., et al. 2000. J Biol Chem. 275: 32775-82. PMID: 10922363
- Purification and characterization of a glutathione S-transferase from the fungus Cunninghamella elegans. | Cha, CJ., et al. 2001. FEMS Microbiol Lett. 203: 257-61. PMID: 11583857
- An ensemble of theta class glutathione transferases with novel catalytic properties generated by stochastic recombination of fragments of two mammalian enzymes. | Broo, K., et al. 2002. J Mol Biol. 318: 59-70. PMID: 12054768
- Non-active site residues Cys69 and Asp150 affected the enzymatic properties of glutathione S-transferase AdGSTD3-3. | Wongtrakul, J., et al. 2003. Insect Biochem Mol Biol. 33: 971-9. PMID: 14505690
- Intra-subunit residue interactions from the protein surface to the active site of glutathione S-transferase AdGSTD3-3 impact on structure and enzyme properties. | Wongtrakul, J., et al. 2005. Insect Biochem Mol Biol. 35: 197-205. PMID: 15705499
- Residue 234 is a master switch of the alternative-substrate activity profile of human and rodent theta class glutathione transferase T1-1. | Shokeer, A. and Mannervik, B. 2010. Biochim Biophys Acta. 1800: 466-73. PMID: 20097269
- Molecular evolution of Theta-class glutathione transferase for enhanced activity with the anticancer drug 1,3-bis-(2-chloroethyl)-1-nitrosourea and other alkylating agents. | Larsson, AK., et al. 2010. Arch Biochem Biophys. 497: 28-34. PMID: 20211594
- Glutathione-binding site of a bombyx mori theta-class glutathione transferase. | Hossain, MD., et al. 2014. PLoS One. 9: e97740. PMID: 24848539
- Identification and functional characterization of an epsilon glutathione S-transferase from the beet armyworm (Spodoptera exigua). | Wan, H., et al. 2016. Pestic Biochem Physiol. 132: 81-8. PMID: 27521917
- Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. | Habig, WH., et al. 1974. J Biol Chem. 249: 7130-9. PMID: 4436300
- Mercapturic acid formation: the several glutathione transferases of rat liver. | Pabst, MJ., et al. 1973. Biochem Biophys Res Commun. 52: 1123-8. PMID: 4736908
- Isoelectric focusing of glutathione S-transferases from rat liver and kidney. | Hales, BF., et al. 1978. Biochem J. 175: 937-43. PMID: 743240
- Evidence that human class Theta glutathione S-transferase T1-1 can catalyse the activation of dichloromethane, a liver and lung carcinogen in the mouse. Comparison of the tissue distribution of GST T1-1 with that of classes Alpha, Mu and Pi GST in human. | Sherratt, PJ., et al. 1997. Biochem J. 326 (Pt 3): 837-46. PMID: 9307035
Activator of:
GSTT1.Substrate of:
GSTT1.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Nitrophenethyl bromide, 5 g | sc-238931A | 5 g | $48.00 | |||
4-Nitrophenethyl bromide, 25 g | sc-238931 | 25 g | $152.00 |
