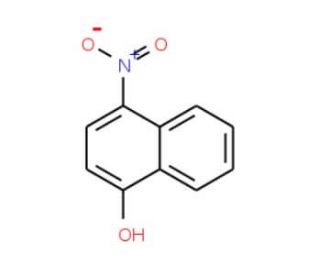
Molecular structure of 4-Nitro-1-naphthol, CAS Number: 605-62-9
4-Nitro-1-naphthol (CAS 605-62-9)
CAS Number:
605-62-9
Molecular Weight:
189.17
Molecular Formula:
C10H7NO3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Nitro-1-naphthol, also known as 4-nitronaphthol or 4-NN, is classified as an organic compound within the naphthol family. Within the body, 4-Nitro-1-naphthol is considered an electrophile, capable of forming a covalent bond with numerous nucleophilic sites present in biological molecules, including proteins, DNA, and RNA. This interaction gives rise to a diverse range of effects on various biochemical processes, contingent upon the specific molecule it associates with. By establishing a covalent bond, 4-Nitro-1-naphthol can either inhibit or activate these processes, depending on the nature of its binding partner.
4-Nitro-1-naphthol (CAS 605-62-9) References
- Discovery and synthesis of hydronaphthoquinones as novel proteasome inhibitors. | Ge, Y., et al. 2012. J Med Chem. 55: 1978-98. PMID: 22220566
- Chromogenic substrate from 4-nitro-1-naphthol for hydrolytic enzyme of neutral or slightly acidic optimum pH: 4-nitro-1-naphthyl-β-D-galactopyranoside as an example. | Dang, J., et al. 2013. Bioorg Med Chem Lett. 23: 646-9. PMID: 23267768
- Metal nanocrystal/metal-organic framework core/shell nanostructure from selective self-assembly induced by localization of metal ion precursors on nanocrystal surface. | Ohhashi, T., et al. 2015. J Colloid Interface Sci. 451: 212-5. PMID: 25898115
- Comparison of Candidate Pairs of Hydrolytic Enzymes for Spectrophotometric-dual-enzyme-simultaneous-assay. | Liu, H., et al. 2015. Anal Sci. 31: 421-7. PMID: 25958872
- Effects of humic acid and heavy metals on the sorption of polar and apolar organic pollutants onto biochars. | Wang, F., et al. 2017. Environ Pollut. 231: 229-236. PMID: 28802992
- Direct observation of stepwise intermolecular proton and hydrogen transfer between alcohols and the triplet state of 4-nitro-1-naphthol. | Zhang, F., et al. 2018. Phys Chem Chem Phys. 20: 11876-11881. PMID: 29662995
- Rapid and Sensitive Detection of Aspergillus niger Using a Single-Mediator System Combined with Redox Cycling. | Kwon, J., et al. 2018. Anal Chem. 90: 13491-13497. PMID: 30403470
- Synthesis, pharmacological evaluation and structure-activity relationship of recently discovered enzyme antagonist azoles. | Dorababu, A. 2020. Heliyon. 6: e03656. PMID: 32274429
- Matrix-dependent a/x pair and overdegraded w/y/z ions generated by radical-directed dissociation of peptide radical cations [M]+ in matrix-assisted laser desorption/ionization in-source decay. | Miyazawa, K. and Takayama, M. 2020. J Mass Spectrom. 55: e4668. PMID: 33135249
- Excited state proton transfer of triplet state p-nitrophenylphenol to amine and alcohol: a spectroscopic and kinetic study. | Pan, X., et al. 2022. Phys Chem Chem Phys. 24: 18427-18434. PMID: 35881619
- Complete and selective nitration of tyrosine residue in peptides caused by ultraviolet matrix-assisted laser desorption/ionization. | Takayama, M. 2023. Photochem Photobiol Sci. 22: 687-692. PMID: 36352303
- Synthesis, Chiral Resolution and Enantiomers Absolute Configuration of 4-Nitropropranolol and 7-Nitropropranolol. | Sparaco, R., et al. 2022. Molecules. 28: PMID: 36615252
- Synthesis of (heterocyclicamino) aminoalkylnaphthols and reduced tetrahydro derivatives for possible antimalarial activity. | Nabih, I., et al. 1972. J Pharm Sci. 61: 1500-2. PMID: 5068966
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Nitro-1-naphthol, 5 g | sc-277576 | 5 g | $200.00 |
