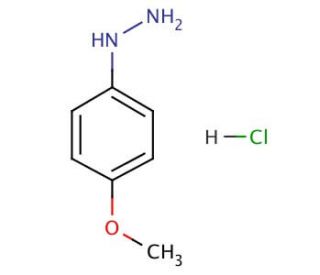
4-Methoxyphenylhydrazine hydrochloride (CAS 19501-58-7)
QUICK LINKS
4-Methoxyphenylhydrazine hydrochloride is a versatile compound used predominantly in the synthesis and modification of various organic molecules, particularly in the field of organic chemistry research. This chemical serves as a hydrazine derivative, where the methoxy group attached to the benzene ring enhances its electronic properties, making it a reagent for specific nucleophilic substitution reactions. It acts through the donation of the hydrazino group, which can engage in the formation of hydrazones and azo compounds, pivotal in the development of dyes, pigments, and other functional materials. The inclusion of the hydrochloride group improves the solubility of this compound in polar solvents, thereby broadening its applicability in different chemical environments. This characteristic is particularly beneficial in facilitating reactions under controlled conditions, thus ensuring greater yield and specificity. 4-Methoxyphenylhydrazine hydrochloride′s role in generating intermediates for further chemical transformations is critical for researchers aiming to design novel compounds with potential applications in materials science and synthetic chemistry.
4-Methoxyphenylhydrazine hydrochloride (CAS 19501-58-7) References
- Reaction of 3-acetonyl-5-cyano-1,2,4-thiadiazole with phenylhydrazine hydrochlorides: indolization and phenylpyrazolation. | Iwakawa, T., et al. 2000. Chem Pharm Bull (Tokyo). 48: 160-2. PMID: 10705497
- An expedient route to the Calabar bean alkaloids (-)-physovenine and (-)-physostigmine. | ElAzab, AS., et al. 2000. Org Lett. 2: 2757-9. PMID: 10964358
- Pyrazole ligands: structure-affinity/activity relationships and estrogen receptor-alpha-selective agonists. | Stauffer, SR., et al. 2000. J Med Chem. 43: 4934-47. PMID: 11150164
- Estrogen pyrazoles: defining the pyrazole core structure and the orientation of substituents in the ligand binding pocket of the estrogen receptor. | Stauffer, SR., et al. 2001. Bioorg Med Chem. 9: 141-50. PMID: 11197334
- Synthesis, characterization of novel coupling products and 4-arylhydrazono-2-pyrazoline-5-ones as potential antimycobacterial agents. | Küçükgüzel, SG. and Rollas, S. 2002. Farmaco. 57: 583-7. PMID: 12164219
- One-pot sequential synthesis of acetoxylated [60]fullerene derivatives. | Chen, ZX. and Wang, GW. 2005. J Org Chem. 70: 2380-3. PMID: 15760237
- Phenolic hydrazones are potent inhibitors of macrophage migration inhibitory factor proinflammatory activity and survival improving agents in sepsis. | Dabideen, DR., et al. 2007. J Med Chem. 50: 1993-7. PMID: 17385848
- Synthesis of novel nitro-substituted triaryl pyrazole derivatives as potential estrogen receptor ligands. | Naoum, F., et al. 2007. Molecules. 12: 1259-73. PMID: 17909483
- Synthesis and exploration of novel curcumin analogues as anti-malarial agents. | Mishra, S., et al. 2008. Bioorg Med Chem. 16: 2894-902. PMID: 18194869
- Synthesis of functionalized tetrasubstituted pyrazolyl heterocycles--a review. | Dadiboyena, S. and Nefzi, A. 2011. Eur J Med Chem. 46: 5258-75. PMID: 21978837
- Preparation of Indole Containing Building Blocks for the Regiospecific Construction of Indole Appended Pyrazoles and Pyrroles. | Gupton, JT., et al. 2013. Tetrahedron. 69: 5829-5840. PMID: 23894213
- Direct arylation of pyridines without the use of a transition metal catalyst. | Li, Y., et al. 2014. Chem Commun (Camb). 50: 7124-7. PMID: 24852113
- Synthesis of Aryl Iodides from Arylhydrazines and Iodine. | Dong, CP., et al. 2018. ACS Omega. 3: 9814-9821. PMID: 31459110
- Synthesis and Characterization of 5-MeO-DMT Succinate for Clinical Use. | Sherwood, AM., et al. 2020. ACS Omega. 5: 32067-32075. PMID: 33344861
- Anhydrous Hydrogen Iodide-Mediated Reductive Indolization of In Situ-Generated Cyclopropyl Hydrazones. | Yasui, M., et al. 2022. Org Lett. 24: 43-47. PMID: 34881907
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Methoxyphenylhydrazine hydrochloride, 10 g | sc-256792 | 10 g | $166.00 |
