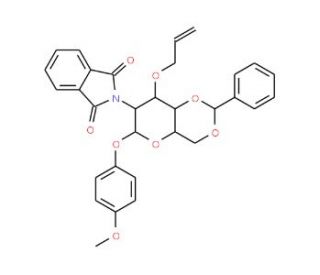
4-Methoxyphenyl 3-O-Allyl-4,6-O-benzylidene-2-deoxy-2-phthalimido-beta-D-glucopyranoside (CAS 889453-84-3)
QUICK LINKS
4-Methoxyphenyl 3-O-Allyl-4,6-O-benzylidene-2-deoxy-2-phthalimido-beta-D-glucopyranoside is a synthetic carbohydrate derivative with an complex structure, primarily utilized in glycosylation research. The inclusion of the phthalimido group at the 2-deoxy position offers a way to study glycosylation processes involving nitrogen functionalities, mimicking amino sugars such as glucosamine. The benzylidene group acts as a protective unit, providing regioselectivity while allowing controlled exposure of hydroxyl groups for subsequent modification. The allyl group offers additional reactivity for conjugation or elongation reactions. This compound plays a critical role as a glycosyl donor, enabling selective glycosylation in oligosaccharide synthesis. Its phthalimido and benzylidene protections are crucial in managing complex carbohydrate syntheses, facilitating the exploration of glycan structures and interactions. In research, it has been leveraged to create glycan arrays for studying carbohydrate-protein interactions or developing carbohydrate-based probes. Furthermore, the compound is a versatile intermediate in assembling more complex glycoconjugates, enhancing our understanding of glycobiology. It aids in elucidating the role of specific glycan motifs in cellular recognition processes, including host-pathogen interactions and glycosylation patterns relevant to different signaling pathways. Overall, it is an essential tool in chemical biology research, underpinning the development of novel glycan-based assays and understanding of glycosylation.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Methoxyphenyl 3-O-Allyl-4,6-O-benzylidene-2-deoxy-2-phthalimido-beta-D-glucopyranoside, 1 g | sc-290402 | 1 g | $172.00 | |||
4-Methoxyphenyl 3-O-Allyl-4,6-O-benzylidene-2-deoxy-2-phthalimido-beta-D-glucopyranoside, 5 g | sc-290402A | 5 g | $533.00 |
