4-Iodobenzophenone (CAS 6136-66-9)
QUICK LINKS
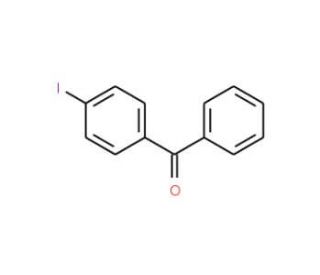
4-Iodobenzophenone (4-IBP) is an organic compound characterized by a benzene ring with an iodine atom attached to the fourth carbon position. Exhibiting a white crystalline solid form, it displays solubility in polar organic solvents like alcohols, ethers, and chloroform. It finds utility in polymer and dye synthesis, as well as in the production of photographic materials. The synthesis of various organic compounds involving 4-Iodobenzophenone often follows a specific reaction pathway. When reacted with a base like potassium carbonate, 4-Iodobenzophenone undergoes a transformation, resulting in the formation of an intermediate compound known as 4-iodobenzoyl chloride. Subsequently, this intermediate reacts with an amine, such as methylamine, to yield the desired end product.
4-Iodobenzophenone (CAS 6136-66-9) References
- An improved method for the palladium-catalyzed amination of aryl iodides. | Ali, MH. and Buchwald, SL. 2001. J Org Chem. 66: 2560-5. PMID: 11304171
- Application of a new bicyclic triaminophosphine ligand in Pd-catalyzed Buchwald-Hartwig amination reactions of aryl chlorides, bromides, and iodides. | Urgaonkar, S., et al. 2003. J Org Chem. 68: 8416-23. PMID: 14575466
- Discovery of leukotriene A4 hydrolase inhibitors using metabolomics biased fragment crystallography. | Davies, DR., et al. 2009. J Med Chem. 52: 4694-715. PMID: 19618939
- Designed synthesis of multi-electrochromic systems bearing diaryl ketone and isophthalates. | Sharmoukh, W., et al. 2010. J Org Chem. 75: 6708-11. PMID: 20828180
- A recyclable Au(I) catalyst for selective homocoupling of arylboronic acids: significant enhancement of nano-surface binding for stability and catalytic activity. | Zhang, X., et al. 2010. J Nanosci Nanotechnol. 10: 5153-60. PMID: 21125864
- Selective Co/Ti cooperatively catalyzed biaryl couplings of aryl halides with aryl metal reagents. | Zeng, J., et al. 2013. Org Lett. 15: 5342-5. PMID: 24093604
- Optimization and Evaluation of 5-Styryl-Oxathiazol-2-one Mycobacterium tuberculosis Proteasome Inhibitors as Potential Antitubercular Agents. | Russo, F., et al. 2015. ChemistryOpen. 4: 342-62. PMID: 26246997
- Amination-Oxidation Strategy for the Copper-Catalyzed Synthesis of Monoarylamines. | Thomas, C., et al. 2016. J Org Chem. 81: 330-5. PMID: 26652145
- Preparation of Polyfunctional Arylzinc Organometallics in Toluene by Halogen/Zinc Exchange Reactions. | Balkenhohl, M., et al. 2019. Angew Chem Int Ed Engl. 58: 12898-12902. PMID: 31243862
- Visible-Light-Driven Thioesterification of Aryl Halides with Potassium Thiocarboxylates: Transition-Metal Catalyst-Free Incorporation of Sulfur Functionalities into an Aromatic Ring. | Volkov, AA., et al. 2022. J Org Chem. 87: 8170-8182. PMID: 35653579
- 3 alpha-Hydroxy-3 beta-(phenylethynyl)-5 beta-pregnan-20-ones: synthesis and pharmacological activity of neuroactive steroids with high affinity for GABAA receptors. | Upasani, RB., et al. 1997. J Med Chem. 40: 73-84. PMID: 9016330
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Iodobenzophenone, 1 g | sc-284261 | 1 g | $73.00 | |||
4-Iodobenzophenone, 5 g | sc-284261A | 5 g | $296.00 |
