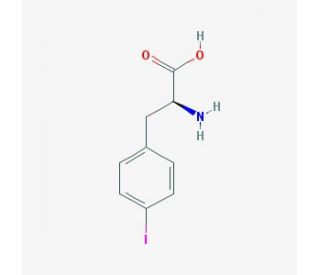
Molecular structure of 4-Iodo-L-phenylalanine, CAS Number: 24250-85-9
4-Iodo-L-phenylalanine (CAS 24250-85-9)
Alternate Names:
p-Iodo-L-phenylalanine
Application:
4-Iodo-L-phenylalanine is used in protein engineering as a model unnatural α amino acid to alter primary amino acid composition via the opal (UGA) codon
CAS Number:
24250-85-9
Purity:
≥98%
Molecular Weight:
291.10
Molecular Formula:
C9H10INO2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Iodo-L-phenylalanine may be used in protein engineering as a model unnatural alpha amino acid to alter primary amino acid composition via the opal (UGA) codon. The presence of 4-Iodo-L-phenylalanine inhibits translation by interacting with the ribosome during the translation process. Additionally, it binds to the hydroxyl group on an mRNA molecule during transcription. As a result of this inhibition, there is a decrease in protein synthesis and the proliferation of cells.
4-Iodo-L-phenylalanine (CAS 24250-85-9) References
- p-ethynylphenylalanine: a potent inhibitor of tryptophan hydroxylase. | Stokes, AH., et al. 2000. J Neurochem. 74: 2067-73. PMID: 10800950
- Regioselective carbon-carbon bond formation in proteins with palladium catalysis; new protein chemistry by organometallic chemistry. | Kodama, K., et al. 2006. Chembiochem. 7: 134-9. PMID: 16307466
- A new protein engineering approach combining chemistry and biology, part I; site-specific incorporation of 4-iodo-L-phenylalanine in vitro by using misacylated suppressor tRNAPhe. | Kodama, K., et al. 2006. Chembiochem. 7: 1577-81. PMID: 16969782
- Site-specific functionalization of proteins by organopalladium reactions. | Kodama, K., et al. 2007. Chembiochem. 8: 232-8. PMID: 17195252
- Site-specific incorporation of 4-iodo-L-phenylalanine through opal suppression. | Kodama, K., et al. 2010. J Biochem. 148: 179-87. PMID: 20495012
- Differentiation of enantiomeric drugs by iodo-substituted L-amino acid references under electrospray ionization mass spectrometric conditions. | Karthikraj, R., et al. 2012. Rapid Commun Mass Spectrom. 26: 1385-91. PMID: 22555933
- Enantiomeric differentiation of β-amino alcohols under electrospray ionization mass spectrometric conditions. | Karthikraj, R., et al. 2014. J Mass Spectrom. 49: 108-16. PMID: 24446270
- Radiopaque, iodine functionalized, phenylalanine-based poly(ester urea)s. | Li, S., et al. 2015. Biomacromolecules. 16: 615-24. PMID: 25575022
- Site-directed spin labelling of proteins by Suzuki-Miyaura coupling via a genetically encoded aryliodide amino acid. | Kugele, A., et al. 2019. Chem Commun (Camb). 55: 1923-1926. PMID: 30680379
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Iodo-L-phenylalanine, 1 g | sc-216895 | 1 g | $143.00 | |||
4-Iodo-L-phenylalanine, 5 g | sc-216895A | 5 g | $275.00 | |||
4-Iodo-L-phenylalanine, 25 g | sc-216895B | 25 g | $541.00 |
