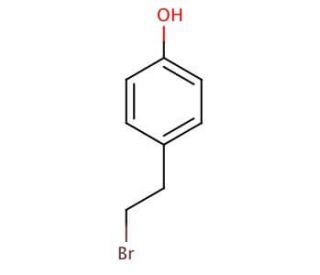
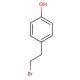
4-Hydroxyphenethyl bromide (CAS 14140-15-9)
QUICK LINKS
4-Hydroxyphenethyl bromide is a chemical compound that functions as an alkylating agent. Its mechanism of action involves the alkylation of nucleophilic groups in biomolecules, such as DNA and proteins, leading to the formation of covalent bonds with these molecules. 4-Hydroxyphenethyl Bromide′s ability to alkylate biomolecules can be utilized to study the effects of covalent modifications on the structure and function of biological macromolecules. 4-Hydroxyphenethyl bromide can be used to investigate the impact of alkylation on cellular processes and signaling pathways, providing insights into the molecular mechanisms underlying various biological phenomena. Its role as an alkylating agent allows for the exploration of the consequences of covalent modifications in a controlled experimental environment, contributing to the understanding of biochemical and cellular processes at the molecular level.
4-Hydroxyphenethyl bromide (CAS 14140-15-9) References
- Structure-activity relationships and cancer-cell selective toxicity of novel inhibitors of glioma-associated oncogene homologue 1 (Gli1) mediated transcription. | Mahindroo, N., et al. 2009. J Med Chem. 52: 4277-87. PMID: 19545120
- Noninvasive Urine Biomarker Lateral Flow Immunoassay for Monitoring Active Onchocerciasis. | Shirey, RJ., et al. 2018. ACS Infect Dis. 4: 1423-1431. PMID: 30141624
- Development of new HO-1 inhibitors by a thorough scaffold-hopping analysis. | Floresta, G., et al. 2018. Bioorg Chem. 81: 334-339. PMID: 30189413
- Progress on the development of floating photobioreactor for microalgae cultivation and its application potential. | Zhu, C., et al. 2019. World J Microbiol Biotechnol. 35: 190. PMID: 31754912
- Synthesis and Evaluation of Artemisinin-Based Hybrid and Dimer Derivatives as Antimelanoma Agents. | Botta, L., et al. 2020. ACS Omega. 5: 243-251. PMID: 31956771
- New Arylethanolimidazole Derivatives as HO-1 Inhibitors with Cytotoxicity against MCF-7 Breast Cancer Cells. | Ciaffaglione, V., et al. 2020. Int J Mol Sci. 21: PMID: 32168943
- C(1)-Phenethyl Derivatives of [closo-1-CB11 H12 ]- and [closo-1-CB9 H10 ]- Anions: Difunctional Building Blocks for Molecular Materials. | Jakubowski, R., et al. 2020. Chemistry. 26: 17481-17494. PMID: 32776629
- Enhancing Microalgal Biomass Productivity in Floating Photobioreactors with Semi-Permeable Membranes Grafted with 4-Hydroxyphenethyl Bromide | , et al. (2020). Macromolecular Research. volume 28,: pages 145–151.
- Extended-release of opioids using fentanyl-based polymeric nanoparticles for enhanced pain management† | Marina Kovaliovab, Shaohua Liab, Emrullah Korkmazc, Devora Cohen-Karniab, Nestor Tomyczb, O. Burak Ozdoganlarcde and Saadyah AverickORCID logo*ab. 2017,. RSC Adv.,. 7,: 47904-47912.
- Noninvasive Urine Biomarker Lateral Flow Immunoassay for Monitoring Active Onchocerciasis | Ryan J. Shirey, Daniel Globisch, Lisa M. Eubanks, Mark S. Hixon, and Kim D. Janda*. 2018. ACS Infect. Dis., 4, 10,: 1423–1431.
- Synthesis of linear polymers containing benzoxazine moieties in the main chain with high molecular design versatility via click reaction | A Chernykh, T Agag, H Ishida - Polymer, 2009 - Elsevier. 16 January 2009,. Polymer. Volume 50, Issue 2,: Pages 382-390.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Hydroxyphenethyl bromide, 1 g | sc-226686 | 1 g | $50.00 |
