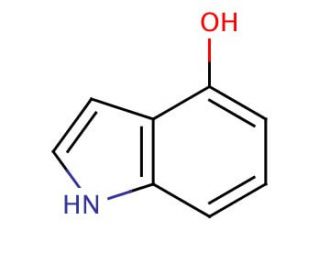
Molecular structure of 4-Hydroxyindole, CAS Number: 2380-94-1
4-Hydroxyindole (CAS 2380-94-1)
Application:
4-Hydroxyindole is a pindolol impurity
CAS Number:
2380-94-1
Purity:
≥98%
Molecular Weight:
133.15
Molecular Formula:
C8H7NO
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Hydroxyindole (4-HI) is an aromatic heterocyclic compound that garners significant attention in scientific research and laboratory experiments. This naturally occurring compound is found abundantly in plants, fungi, and animals and serves as a component in the production of various industrial products. While the precise mechanism of action of 4-Hydroxyindole remains under investigation, it is believed to exert its effects by inhibiting specific enzymes, including cytochrome P450 and monoamine oxidase.
4-Hydroxyindole (CAS 2380-94-1) References
- Conversion of 1-benzoylindole by Aspergillus strains. | Sukhodolskaya, GV., et al. 2000. Appl Microbiol Biotechnol. 53: 695-700. PMID: 10919329
- Rational design of an indolebutanoic acid derivative as a novel aldose reductase inhibitor based on docking and 3D QSAR studies of phenethylamine derivatives. | Sun, WS., et al. 2003. J Med Chem. 46: 5619-27. PMID: 14667216
- Inhibition of amyloid fibril formation and cytotoxicity by hydroxyindole derivatives. | Cohen, T., et al. 2006. Biochemistry. 45: 4727-35. PMID: 16605241
- Synthesis and biological evaluation of novel angular fused Pyrrolocoumarins. | Kontogiorgis, C., et al. 2008. J Enzyme Inhib Med Chem. 23: 43-9. PMID: 18341252
- Glucuronidation of psilocin and 4-hydroxyindole by the human UDP-glucuronosyltransferases. | Manevski, N., et al. 2010. Drug Metab Dispos. 38: 386-95. PMID: 20007669
- Bicyclic compounds repress membrane vesicle production and Pseudomonas quinolone signal synthesis in Pseudomonas aeruginosa. | Tashiro, Y., et al. 2010. FEMS Microbiol Lett. 304: 123-30. PMID: 20146747
- In vitro assay of six UDP-glucuronosyltransferase isoforms in human liver microsomes, using cocktails of probe substrates and liquid chromatography-tandem mass spectrometry. | Seo, KA., et al. 2014. Drug Metab Dispos. 42: 1803-10. PMID: 25122565
- Biocatalytic Production of Psilocybin and Derivatives in Tryptophan Synthase-Enhanced Reactions. | Blei, F., et al. 2018. Chemistry. 24: 10028-10031. PMID: 29750381
- Biofilm Formation by Streptococcus mutans is Enhanced by Indole via the Quorum Sensing Pathway. | Inaba, T., et al. 2020. Microbes Environ. 35: PMID: 32350164
- Synthetic studies toward inducamide C. | Nabi, AA., et al. 2021. Org Biomol Chem. 19: 416-420. PMID: 33313627
- Development and validation of an LC-MS/MS method for the bioanalysis of psilocybin's main metabolites, psilocin and 4-hydroxyindole-3-acetic acid, in human plasma. | Kolaczynska, KE., et al. 2021. J Chromatogr B Analyt Technol Biomed Life Sci. 1164: 122486. PMID: 33485158
- Fluorescent Pyranoindole Congeners: Synthesis and Photophysical Properties of Pyrano[3,2-f], [2,3-g], [2,3-f], and [2,3-e]Indoles. | Sharapov, AD., et al. 2022. Molecules. 27: PMID: 36557999
- Psammopemmins (A-C), Novel Brominated 4-Hydroxyindole Alkaloids From an Antarctic Sponge, Psammopemma sp. | MS Butler, RJ Capon and CC Lu. 1871 - 1877. Australian Journal of Chemistry. 45(11): 1871 - 1877.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Hydroxyindole, 1 g | sc-216890 | 1 g | $215.00 |
