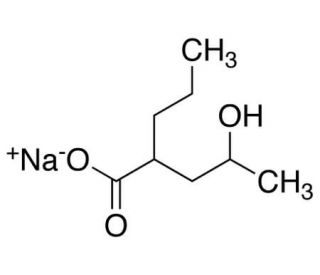
4-Hydroxy Valproic Acid Sodium Salt (CAS 1216888-06-0)
QUICK LINKS
4-Hydroxy Valproic Acid Sodium Salt(Mixture of Diastereomers) is a racemic mixture of metabolites of Valproic Acid. In the metabolism of Valproic Acid to 4-Hydroxy Valproic Acid, it has been reported that CYP4B1 may play a role in this conversion, as demonstrated using monospecific polyclonal antibodies directed against CYP4B1. Valproic Acid is a branched chain, fatty acid which is reported to potentially enhance central GABAergic neurotransmission and inhibit Na+ channels. Currently, the various molecular mechanisms of action of Valproic Acid have not been completely elucidated. Valproic Acid has been reported to be a potent inhibitor of histone deacetylases (HDACs) in vitro. Valproic Acid is also noted to relieve HDAC-dependent transcriptional repression and causes the hyperacetylation of histones in cultured cells. In animal studies, Valproic Acid has been observed to reduce tumor growth and metastasis formation. Additionally, Valproic Acid is reported to activate Wnt-dependent gene expression and to mimic trichostatin A (sc-3511) in the inhibition of histone deacetylase.
Metabolites of Valproic Acid are also available as:
Valproic Acid (sc-213144)
Valproic Acid, Sodium Salt (sc-202378)
3-Hydroxy Valproic Acid (sc-209599)
3-Keto Valproic Acid Sodium Salt (sc-216476)
rac 5-Hydroxy Valproic Acid Sodium Salt (sc-208266)
Valproic Acid β-D-Glucuronide Allyl Ester (sc-220360)
Valproic Acid β-D-Glucuronide (sc-213145)
Labelled forms of Valproic Acid are available as:
Valproic Acid-d6 (sc-213146)
Valproic Acid-d6 β-D-Glucuronide (sc-220361)
rac 5-Hydroxy Valproic Acid-d7 Sodium Salt (sc-219789)
4-Hydroxy Valproic Acid Sodium Salt (CAS 1216888-06-0) References
- Do structural properties explain the anticonvulsant activity of valproate metabolites? A QSAR analysis. | Bello-Ramírez, AM., et al. 2002. Epilepsia. 43: 475-81. PMID: 12027907
- Studies on the beta-oxidation of valproic acid in rat liver mitochondrial preparations. | Bjorge, SM. and Baillie, TA. 1991. Drug Metab Dispos. 19: 823-9. PMID: 1680661
- Development of human liver UDP-glucuronosyltransferases. | Burchell, B., et al. 1989. Dev Pharmacol Ther. 13: 70-7. PMID: 2515047
- Determination of valproic acid and its six metabolites in human serum using LC-MS/MS and application to interaction with carbapenems in epileptic patients. | Yang, X., et al. 2023. Biomed Chromatogr. 37: e5572. PMID: 36520520
- Successful LC-MS/MS assay development and validation for determination of valproic acid and its metabolites supporting proactive pharmacovigilance. | Wang, WJ., et al. 2023. J Pharm Biomed Anal. 234: 115538. PMID: 37354631
- CYP4 isozyme specificity and the relationship between omega-hydroxylation and terminal desaturation of valproic acid. | Rettie, AE., et al. 1995. Biochemistry. 34: 7889-95. PMID: 7794900
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Hydroxy Valproic Acid Sodium Salt, 100 mg | sc-216870 | 100 mg | $330.00 |
