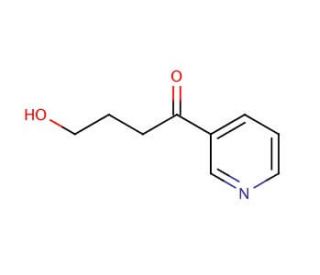
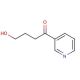
Molecular structure of 4-Hydroxy-1-(3-pyridyl)-1-butanone, CAS Number: 59578-62-0
4-Hydroxy-1-(3-pyridyl)-1-butanone (CAS 59578-62-0)
Application:
4-Hydroxy-1-(3-pyridyl)-1-butanone is a metabolite of NNK
CAS Number:
59578-62-0
Molecular Weight:
165.19
Molecular Formula:
C9H11NO2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Hydroxy-1-(3-pyridyl)-1-butanone is an analog of nicotinic acid with a hydroxy butyl ketone substituted for the carboxylic acid. 4-Hydroxy-1-(3-pyridyl)-1-butanone is useful in the study of tobacco-specific nitrosamine compounds and is relevant to studying the metabolism pathway of the potent tobacco-specific mutagen 4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanone (sc-209854), also known as NNK.
4-Hydroxy-1-(3-pyridyl)-1-butanone (CAS 59578-62-0) References
- The pyridyloxobutyl DNA adduct, O6-[4-oxo-4-(3-pyridyl)butyl]guanine, is detected in tissues from 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone-treated A/J mice. | Thomson, NM., et al. 2003. Chem Res Toxicol. 16: 1-6. PMID: 12693024
- Metabolism of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone in human lung and liver microsomes and cytochromes P-450 expressed in hepatoma cells. | Smith, TJ., et al. 1992. Cancer Res. 52: 1757-63. PMID: 1312898
- Investigation of the reaction of myosmine with sodium nitrite in vitro and in rats. | Hecht, SS., et al. 2007. Chem Res Toxicol. 20: 543-9. PMID: 17291014
- Synthesis of 4-[2-aminoethyl(nitrosamino)]-1-pyridin-3-yl-butan-1-one, a new NNK hapten for the induction of N-nitrosamine-specific antibodies. | Prodhomme, EJ., et al. 2007. Bioconjug Chem. 18: 2045-53. PMID: 17939731
- Detection and quantification of 4-hydroxy-1-(3-pyridyl)-1-butanone (HPB) from smoker albumin and its potential as a surrogate biomarker of tobacco-specific nitrosamines exposure and bioactivation. | Wang, Y., et al. 2019. Toxicol Lett. 311: 11-16. PMID: 31026483
- Synergistic Effect between Human Papillomavirus 18 and 4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanone on Malignant Transformation of Immortalized SHEE Cells. | Zhuang, Z., et al. 2020. Chem Res Toxicol. 33: 470-481. PMID: 31874558
- Formation of hemoglobin adducts upon treatment of F344 rats with the tobacco-specific nitrosamines 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone and N'-nitrosonornicotine. | Carmella, SG. and Hecht, SS. 1987. Cancer Res. 47: 2626-30. PMID: 3567895
- Evidence that a hemoglobin adduct used for dosimetry of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone is a carboxylic ester. | Carmella, SG., et al. 1993. Environ Health Perspect. 99: 203-5. PMID: 8319624
- Simultaneous solid-phase extraction and gas chromatographic-mass spectrometric determination of hemoglobin adducts from tobacco-specific nitrosamines and aromatic amines. | Kutzer, C., et al. 1997. J Chromatogr Sci. 35: 1-6. PMID: 8989869
- Pyridyloxobutyl adduct O6-[4-oxo-4-(3-pyridyl)butyl]guanine is present in 4-(acetoxymethylnitrosamino)-1-(3-pyridyl)-1-butanone-treated DNA and is a substrate for O6-alkylguanine-DNA alkyltransferase. | Wang, L., et al. 1997. Chem Res Toxicol. 10: 562-7. PMID: 9168254
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Hydroxy-1-(3-pyridyl)-1-butanone, 10 mg | sc-210066 | 10 mg | $360.00 |
