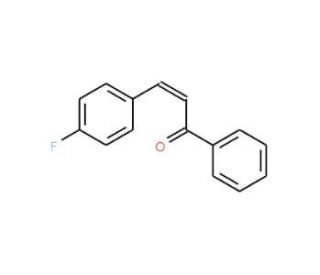
4-Fluorochalcone (CAS 1608-51-1)
QUICK LINKS
4-Fluorochalcone is a compound that functions as a chemical intermediate in organic synthesis. It participates in various reactions to form more complex molecules, serving as a building block for the synthesis of agrochemicals, and other organic compounds. Its mechanism of action involves undergoing nucleophilic addition reactions with various reagents to form chalcone derivatives, which can further undergo cyclization, oxidation, or reduction to yield diverse chemical structures. 4-Fluorochalcone′s role in the synthesis of chalcone derivatives may be versatile for the development of new chemical entities with potential applications in various fields of development. At the molecular level, 4-Fluorochalcone interacts with different reagents and catalysts to facilitate the formation of carbon-carbon and carbon-heteroatom bonds, contributing to the diversification of chemical libraries for screening and evaluation in experimental applications.
4-Fluorochalcone (CAS 1608-51-1) References
- Investigations on the vibrational modes and non-linear optical properties of 4-Fluoro Chalcone crystal. | Prabu, S., et al. 2014. Spectrochim Acta A Mol Biomol Spectrosc. 129: 114-20. PMID: 24727169
- Synthesis and characterization of fluorinated azadipyrromethene complexes as acceptors for organic photovoltaics. | Etheridge, FS., et al. 2016. Beilstein J Org Chem. 12: 1925-1938. PMID: 27829899
- Evaluation of chalcones as inhibitors of glutathione S-transferase. | Özaslan, MS., et al. 2018. J Biochem Mol Toxicol. 32: e22047. PMID: 29473699
- The behavior of some chalcones on acetylcholinesterase and carbonic anhydrase activity. | Aslan, HE., et al. 2019. Drug Chem Toxicol. 42: 634-640. PMID: 29860891
- Mechanisms of Multidrug Resistance in Cancer Chemotherapy. | Bukowski, K., et al. 2020. Int J Mol Sci. 21: PMID: 32370233
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Fluorochalcone, 5 g | sc-284236 | 5 g | $80.00 | |||
4-Fluorochalcone, 25 g | sc-284236A | 25 g | $240.00 |
