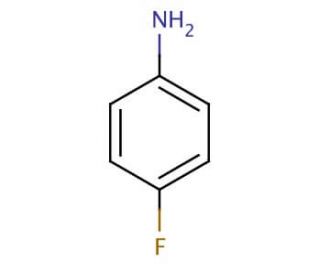
4-Fluoroaniline (CAS 371-40-4)
QUICK LINKS
4-Fluoroaniline is a chemical compound that functions as a building block in the synthesis of various organic compounds. It acts as a precursor in the production of agrochemicals and dyes. 4-Fluoroaniline participates in nucleophilic aromatic substitution reactions, where it undergoes substitution with other functional groups to form new compounds. Its mechanism of action involves its ability to react with electrophiles, leading to the formation of new carbon-carbon or carbon-heteroatom bonds. 4-Fluoroaniline plays a role in the modification of organic molecules, allowing for the creation of diverse chemical structures for further study and potential applications in various industries. At the molecular level, 4-Fluoroaniline′s reactivity and selectivity make it useful for the synthesis of complex organic molecules with specific properties.
4-Fluoroaniline (CAS 371-40-4) References
- Studies on the metabolism of 4-fluoroaniline and 4-fluoroacetanilide in rat: formation of 4-acetamidophenol (paracetamol) and its metabolites via defluorination and N-acetylation. | Scarfe, GB., et al. 1999. Xenobiotica. 29: 205-16. PMID: 10199596
- Metabolism of 4-fluoroaniline and 4-fluorobiphenyl in the earthworm Eisenia veneta characterized by high-resolution NMR spectroscopy with directly coupled HPLC-NMR and HPLC-MS. | Bundy, JG., et al. 2002. Xenobiotica. 32: 479-90. PMID: 12160481
- Metabonomic assessment of toxicity of 4-fluoroaniline, 3,5-difluoroaniline and 2-fluoro-4-methylaniline to the earthworm Eisenia veneta (Rosa): identification of new endogenous biomarkers. | Bundy, JG., et al. 2002. Environ Toxicol Chem. 21: 1966-72. PMID: 12206438
- Potential of neurotoxicity after a single oral dose of 4-bromo-, 4-chloro-, 4-fluoro- or 4-iodoaniline in rats. | Okazaki, Y., et al. 2003. J Appl Toxicol. 23: 315-22. PMID: 12975770
- Bioactivation of 4-fluorinated anilines to benzoquinoneimines as primary reaction products. | Rietjens, IM. and Vervoort, J. 1991. Chem Biol Interact. 77: 263-81. PMID: 2009574
- Differences between 4-fluoroaniline degradation and autoinducer release by Acinetobacter sp. TW: implications for operating conditions in bacterial bioaugmentation. | Wang, M., et al. 2013. Environ Sci Pollut Res Int. 20: 6201-9. PMID: 23589249
- Isolation, identification and characterization of a novel Ralstonia sp. FD-1, capable of degrading 4-fluoroaniline. | Song, E., et al. 2014. Biodegradation. 25: 85-94. PMID: 23604516
- Reactions of 4 methylphenyl isocyanate with amino acids. | Gabriele Sabbioni John H Lamb Peter B Farmer And Ovnair Sepai,. 1997. Biomarkers. 2: 223-32. PMID: 23899214
- Development of an LC-MS Method for 4-Fluoroaniline Determination in Ezetimibe. | Dong, S., et al. 2018. J Chromatogr Sci. 56: 724-730. PMID: 29762651
- General Nondestructive Passivation by 4-Fluoroaniline for Perovskite Solar Cells with Improved Performance and Stability. | Zhao, S., et al. 2018. Small. 14: e1803350. PMID: 30417558
- Aerobic degradation of 4-fluoroaniline and 2,4-difluoroaniline: performance and microbial community in response to the inocula. | Zhao, ZQ., et al. 2021. Biodegradation. 32: 53-71. PMID: 33428058
- Synthesis and Biological Evaluation of Highly Active 7-Anilino Triazolopyrimidines as Potent Antimicrotubule Agents. | Oliva, P., et al. 2022. Pharmaceutics. 14: PMID: 35745764
- Exploring Highly Functionalized Tetrahydropyridine as a Dual Inhibitor of Monoamine Oxidase A and B: Synthesis, Structural Analysis, Single Crystal XRD, Supramolecular Assembly Exploration by Hirshfeld Surface Analysis, and Computational Studies. | Khan, BA., et al. 2022. ACS Omega. 7: 29452-29464. PMID: 36033707
- Microbial mineralization of ring-substituted anilines through an ortho-cleavage pathway. | Zeyer, J., et al. 1985. Appl Environ Microbiol. 50: 447-53. PMID: 4051488
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Fluoroaniline, 25 g | sc-254669 | 25 g | $20.00 | |||
4-Fluoroaniline, 100 g | sc-254669A | 100 g | $36.00 |
