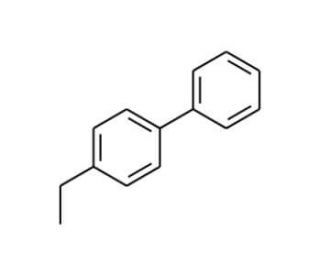
4-Ethylbiphenyl (CAS 5707-44-8)
QUICK LINKS
4-Ethylbiphenyl is a compound that plays a significant role in materials science and organic synthesis. Its biphenyl core structure is important in the study of liquid crystalline materials due to the rigidity and planar nature of the two phenyl rings, which can induce anisotropic physical properties for the operation of liquid crystal displays (LCDs). In organic synthesis, the ethyl group at the 4-position provides a point of chemical reactivity for further functionalization, allowing chemists to utilize it as a building block for more complex molecular structures. It′s in the synthesis of various organic compounds including polymers, where its incorporation can affect the thermal and optical properties of the material. 4-Ethylbiphenyl′s structure is relevant in the field of host-guest chemistry, where it can act as a host compound for the formation of inclusion complexes, potentially useful in the development of new chemical delivery systems.
4-Ethylbiphenyl (CAS 5707-44-8) References
- Influence of dispersants on bioconcentration factors of seven organic compounds with different lipophilicities and structures. | Yakata, N., et al. 2006. Chemosphere. 64: 1885-91. PMID: 16527330
- Kinetics and mechanism of oxygen-independent hydrocarbon hydroxylation by ethylbenzene dehydrogenase. | Szaleniec, M., et al. 2007. Biochemistry. 46: 7637-46. PMID: 17542621
- Porous metalloporphyrinic frameworks constructed from metal 5,10,15,20-tetrakis(3,5-biscarboxylphenyl)porphyrin for highly efficient and selective catalytic oxidation of alkylbenzenes. | Yang, XL., et al. 2012. J Am Chem Soc. 134: 10638-45. PMID: 22650149
- Substrate and inhibitor spectra of ethylbenzene dehydrogenase: perspectives on application potential and catalytic mechanism. | Knack, D., et al. 2012. Appl Environ Microbiol. 78: 6475-82. PMID: 22773630
- Efficient benzylic and aliphatic C-H oxidation with selectivity for methylenic sites catalyzed by a bioinspired manganese complex. | Shen, D., et al. 2014. Org Lett. 16: 1108-11. PMID: 24499500
- Cis-Selective Decarboxylative Alkenylation of Aliphatic Carboxylic Acids with Vinyl Arenes Enabled by Photoredox/Palladium/Uphill Triple Catalysis. | Zheng, C., et al. 2018. Org Lett. 20: 2559-2563. PMID: 29664648
- A comparative study for the organic byproducts from hydrothermal carbonizations of sugarcane bagasse and its bio-refined components cellulose and lignin. | Du, FL., et al. 2018. PLoS One. 13: e0197188. PMID: 29856735
- Nickel-Catalyzed Decarbonylative Alkylation of Aroyl Fluorides Assisted by Lewis-Acidic Organoboranes. | Okuda, Y., et al. 2018. ACS Omega. 3: 13129-13140. PMID: 31458033
- Copper-catalysed benzylic C-H coupling with alcohols via radical relay enabled by redox buffering. | Hu, H., et al. 2020. Nat Catal. 3: 358-367. PMID: 32368720
- Asymmetric benzylic C(sp3)-H acylation via dual nickel and photoredox catalysis. | Huan, L., et al. 2021. Nat Commun. 12: 3536. PMID: 34112783
- Photochemical and Electrochemical Applications of Proton-Coupled Electron Transfer in Organic Synthesis. | Murray, PRD., et al. 2022. Chem Rev. 122: 2017-2291. PMID: 34813277
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Ethylbiphenyl, 5 g | sc-232656 | 5 g | $48.00 |
