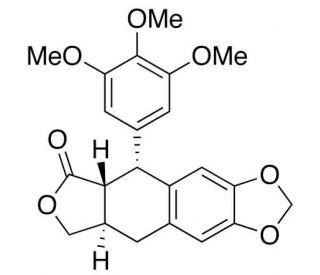
4-Deoxypodophyllotoxin (CAS 19186-35-7)
QUICK LINKS
4-Deoxypodophyllotoxin is an occurring lignan compound that functions as a potent inhibitor of microtubule assembly. It exerts its mechanism of action by binding to the colchicine-binding site on tubulin, thereby disrupting microtubule dynamics and inhibiting cell division. This disruption ultimately leads to cell cycle arrest in the G2/M phase, preventing the formation of the mitotic spindle and resulting in apoptotic cell death. 4-Deoxypodophyllotoxin may induce DNA damage and inhibit topoisomerase II activity, further contributing to its cytotoxic effects. Its ability to interfere with microtubule formation and disrupt key cellular processes may be valuable for studying cell cycle regulation and potential anti-cancer properties in experimental applications.
4-Deoxypodophyllotoxin (CAS 19186-35-7) References
- 4-Aza-2,3-dehydro-4-deoxypodophyllotoxins: simple aza-podophyllotoxin analogues possessing potent cytotoxicity. | Hitotsuyanagi, Y., et al. 2000. Bioorg Med Chem Lett. 10: 315-7. PMID: 10714489
- Plant-based anticancer molecules: a chemical and biological profile of some important leads. | Srivastava, V., et al. 2005. Bioorg Med Chem. 13: 5892-908. PMID: 16129603
- Replacement of the lactone moiety on podophyllotoxin and steganacin analogues with a 1,5-disubstituted 1,2,3-triazole via ruthenium-catalyzed click chemistry. | Imperio, D., et al. 2007. Bioorg Med Chem. 15: 6748-57. PMID: 17765552
- Natural products-based insecticidal agents 5. Design, semisynthesis and insecticidal activity of novel 4'-substituted benzenesulfonate derivatives of 4-deoxypodophyllotoxin against Mythimna separata Walker in vivo. | Xu, H. and Wang, JJ. 2010. Bioorg Med Chem Lett. 20: 2500-2. PMID: 20346661
- Seasonal variations in the deoxypodophyllotoxin content and yield of Anthriscus sylvestris L. (Hoffm.) grown in the field and under controlled conditions. | Hendrawati, O., et al. 2011. J Agric Food Chem. 59: 8132-9. PMID: 21604818
- Synthesis and insecticidal activity of new deoxypodophyllotoxin-based phenazine analogues against Mythimna separata Walker. | Wang, J., et al. 2013. J Agric Food Chem. 61: 6336-43. PMID: 23756712
- RP-HPLC method using one marker for quantification of four podophyllum lignans in medicinal plants. | Lu, N., et al. 2014. J Chromatogr Sci. 52: 514-9. PMID: 23766105
- Synthesis and insecticidal activity of new deoxypodophyllotoxin derivatives modified in the D-ring. | Wang, J., et al. 2014. Bioorg Med Chem Lett. 24: 4542-4545. PMID: 25127871
- Combinatorial Synthesis of A Series of Paeonol-based Phenylsulfonyl hydrazone Derivatives as Insecticidal Agents. | Che, ZP., et al. 2020. Comb Chem High Throughput Screen. 23: 232-238. PMID: 31985371
- Regioselective suprafacial 1, 5-hydrogen shifts in o-quinodimethanes; a route to 4-deoxypodophyllotoxin | Jones, D. W., & Thompson, A. M. 1988. Journal of the Chemical Society, Chemical Communications. (16): 1095-1096.
- Lignans and related phenols. Part 18. The synthesis of quinones from podophyllotoxin and its analogues | Ayres, D. C., & Ritchie, T. J. 1988. Journal of the Chemical Society, Perkin Transactions 1. (9): 2573-2578.
- Design, semisynthesis and insecticidal activity of novel podophyllotoxin derivatives against Brontispa longissima in vivo | Liu, Y. Q., Zhao, Y. L., Yang, L., Zhou, X. W., & Feng, G. 2012. Pesticide biochemistry and physiology. 102(1): 11-18.
- Synthesis of Cytotoxic Isodeoxypodophyllotoxin Analogs | Alizadeh, B. H., Emami, S., Dehghan, G., Foroumadi, A., & Shafiee, A. 2017. Journal of Heterocyclic Chemistry. 54(1): 539-545.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Deoxypodophyllotoxin, 100 mg | sc-500412 | 100 mg | $388.00 |
