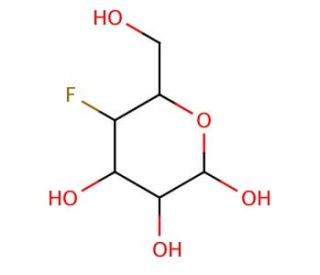
4-Deoxy-4-fluoro-D-glucose (CAS 29218-07-3)
QUICK LINKS
4-Deoxy-4-fluoro-D-glucose, a modified form of glucose with a fluorine atom replacing the hydroxyl group at the C-4 position, serves as a valuable tool in biochemical and metabolic research. Its structural similarity to glucose enables its utilization as a metabolic tracer in various experimental settings. One of the primary research applications of this compound involves its incorporation into cellular metabolic pathways to study glucose metabolism and its impact on cellular functions. Researchers employ 4-Deoxy-4-fluoro-D-glucose in positron emission tomography (PET) imaging studies to investigate glucose uptake and utilization in tissues and organs. By radiolabeling this compound with a positron-emitting isotope such as fluorine-18, scientists can visualize and quantify glucose metabolism in vivo, providing insights into the metabolic activity of healthy and diseased tissues. Additionally, 4-Deoxy-4-fluoro-D-glucose finds applications in elucidating the mechanisms underlying metabolic diseases such as diabetes and cancer. Studies utilizing this compound aim to uncover aberrations in glucose metabolism associated with disease progression and identify potential targets for intervention. Furthermore, its use as a metabolic probe facilitates the development of novel diagnostic tools and strategies for managing metabolic disorders. Through continued research efforts, 4-Deoxy-4-fluoro-D-glucose contributes to advancing our understanding of glucose metabolism and its implications for health and disease.
4-Deoxy-4-fluoro-D-glucose (CAS 29218-07-3) References
- Effects of substituting a OH group by a F atom in D-glucose. Ab initio and DFT analysis. | Hoffmann, M. and Rychlewski, J. 2001. J Am Chem Soc. 123: 2308-16. PMID: 11456879
- A one-pot enzymatic approach to the O-fluoroglucoside of N-methylanthranilate. | Caputi, L., et al. 2013. Bioorg Med Chem. 21: 4762-7. PMID: 23806835
- The identification of perillyl alcohol glycosides with improved antiproliferative activity. | Nandurkar, NS., et al. 2014. J Med Chem. 57: 7478-84. PMID: 25121720
- Anomeric Selectivity of Trehalose Transferase with Rare l-Sugars. | Mestrom, L., et al. 2020. ACS Catal. 10: 8835-8839. PMID: 32953231
- Structures of human SGLT in the occluded state reveal conformational changes during sugar transport. | Cui, W., et al. 2023. Nat Commun. 14: 2920. PMID: 37217492
- Obtaining Pure 1H NMR Spectra of Individual Pyranose and Furanose Anomers of Reducing Deoxyfluorinated Sugars. | Poškaitė, G., et al. 2023. J Org Chem. 88: 13908-13925. PMID: 37754916
- The use of deoxyfluoro-D-galactopyranoses in a study of yeast galactokinase specificity. | Thomas, P., et al. 1974. Biochem J. 139: 661-4. PMID: 4369372
- The use of deoxyfluoro-D-glucopyranoses and related compounds in a study of yeast hexokinase specificity. | Bessell, EM., et al. 1972. Biochem J. 128: 199-204. PMID: 4563639
- An alternative synthesis of 4-deoxy-4-fluoro-D-glucose and its transport in the human erythrocyte. | Lopes, DP. and Taylor, NF. 1979. Carbohydr Res. 73: 125-34. PMID: 476722
- The reaction of 4-deoxy-4-fluoro-D-glucose with an outer membrane protein of Pseudomonas putida. | D'Amore, T. and Taylor, NF. 1982. FEBS Lett. 143: 247-51. PMID: 7117530
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Deoxy-4-fluoro-D-glucose, 25 mg | sc-220935 | 25 mg | $230.00 |
