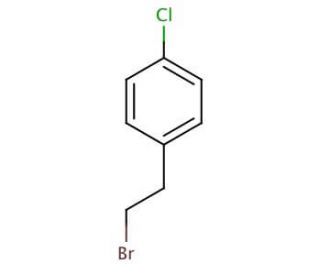
Molecular structure of 4-Chlorophenethyl bromide, CAS Number: 6529-53-9
4-Chlorophenethyl bromide (CAS 6529-53-9)
CAS Number:
6529-53-9
Purity:
≥97%
Molecular Weight:
219.51
Molecular Formula:
C8H8BrCl
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Chlorophenethyl bromide is a versatile compound employed in scientific research applications. Its significance lies in its utility as a reagent for synthesizing diverse organic compounds, including heterocyclic compounds and pesticides. Furthermore, it finds use in the synthesis of polymers, dyes, and various materials. Notably, 4-Chlorophenethyl bromide plays a key role in the synthesis of organometallic compounds, such as organoboranes, and also contributes to the synthesis of organosilicon compounds.
4-Chlorophenethyl bromide (CAS 6529-53-9) References
- The preparation of 3-substituted-1,5-dibromopentanes as precursors to heteracyclohexanes. | Ringstrand, B., et al. 2011. Beilstein J Org Chem. 7: 386-93. PMID: 21512596
- Structure-guided design, synthesis, and evaluation of guanine-derived inhibitors of the eIF4E mRNA-cap interaction. | Chen, X., et al. 2012. J Med Chem. 55: 3837-51. PMID: 22458568
- Discovery of SMP-304, a novel benzylpiperidine derivative with serotonin transporter inhibitory activity and 5-HT1A weak partial agonistic activity showing the antidepressant-like effect. | Yoshinaga, H., et al. 2017. Bioorg Med Chem. 25: 293-304. PMID: 27865645
- An Agonist Radioligand for the Proinflammatory Lipid-Activated G Protein-Coupled Receptor GPR84 Providing Structural Insights. | Köse, M., et al. 2020. J Med Chem. 63: 2391-2410. PMID: 31721581
- Structure-Activity Relationship Studies of α-Ketoamides as Inhibitors of the Phospholipase A and Acyltransferase Enzyme Family. | Zhou, J., et al. 2020. J Med Chem. 63: 9340-9359. PMID: 32787138
- Targeting nuclear protein TDP-43 by cell division cycle kinase 7 inhibitors: A new therapeutic approach for amyotrophic lateral sclerosis. | Rojas-Prats, E., et al. 2021. Eur J Med Chem. 210: 112968. PMID: 33139113
- Design, computational studies, synthesis and in vitro antimicrobial evaluation of benzimidazole based thio-oxadiazole and thio-thiadiazole analogues. | Noureldin, NA., et al. 2021. BMC Chem. 15: 58. PMID: 34711258
- 1-Benzyloxy-5-phenyltetrazole derivatives highly active against androgen receptor-dependent prostate cancer cells. | Zhao, S., et al. 2023. Eur J Med Chem. 246: 114982. PMID: 36495632
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Chlorophenethyl bromide, 5 g | sc-232603 | 5 g | $106.00 |
