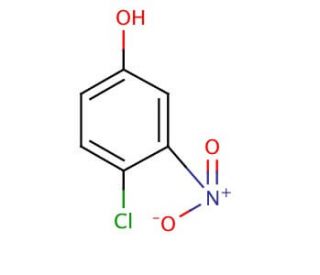
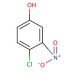
4-Chloro-3-nitrophenol (CAS 610-78-6)
QUICK LINKS
4-Chloro-3-nitrophenol is a chemical compound that functions as a precursor in the synthesis of various organic compounds. It acts as a building block in the production of dyes and agricultural chemicals. The compound′s mode of action involves participating in nucleophilic aromatic substitution reactions, where it undergoes substitution with other functional groups to form new chemical compounds. Role in development is to serve as a starting material for the synthesis of more complex molecules, allowing for the creation of diverse chemical structures for further investigation and potential applications in various industries. At the molecular level, 4-Chloro-3-nitrophenol interacts with other reagents to facilitate the formation of new chemical bonds, enabling the creation of novel compounds with unique properties for experimental exploration.
4-Chloro-3-nitrophenol (CAS 610-78-6) References
- Chemoselective nitro group reduction and reductive dechlorination initiate degradation of 2-chloro-5-nitrophenol by Ralstonia eutropha JMP134. | Schenzle, A., et al. 1999. Appl Environ Microbiol. 65: 2317-23. PMID: 10347008
- QSAR analysis of the toxicity of nitroaromatics in Tetrahymena pyriformis: structural factors and possible modes of action. | Artemenko, AG., et al. 2011. SAR QSAR Environ Res. 22: 575-601. PMID: 21714735
- Genes involved in degradation of para-nitrophenol are differentially arranged in form of non-contiguous gene clusters in Burkholderia sp. strain SJ98. | Vikram, S., et al. 2013. PLoS One. 8: e84766. PMID: 24376843
- Bacterial degradation of chlorophenols and their derivatives. | Arora, PK. and Bae, H. 2014. Microb Cell Fact. 13: 31. PMID: 24589366
- Degradation of 4-chloro-3-nitrophenol via a novel intermediate, 4-chlororesorcinol by Pseudomonas sp. JHN. | Arora, PK., et al. 2014. Sci Rep. 4: 4475. PMID: 24667329
- Evaluating N-benzylgalactonoamidines as putative transition state analogs for β-galactoside hydrolysis. | Fan, QH., et al. 2014. Org Biomol Chem. 12: 2792-800. PMID: 24668069
- Aryl-substituted aminobenzimidazoles targeting the hepatitis C virus internal ribosome entry site. | Ding, K., et al. 2014. Bioorg Med Chem Lett. 24: 3113-7. PMID: 24856063
- Biotransformation and chemotaxis of 4-chloro-2-nitrophenol by Pseudomonas sp. JHN. | Arora, PK. and Bae, H. 2014. Microb Cell Fact. 13: 110. PMID: 25112300
- Genetic and Biochemical Characterization of 2-Chloro-5-Nitrophenol Degradation in a Newly Isolated Bacterium, Cupriavidus sp. Strain CNP-8. | Min, J., et al. 2017. Front Microbiol. 8: 1778. PMID: 28959252
- Recent advances in degradation of chloronitrophenols. | Arora, PK., et al. 2018. Bioresour Technol. 250: 902-909. PMID: 29229201
- Biodegradation of 2-chloro-4-nitrophenol via a hydroxyquinol pathway by a Gram-negative bacterium, Cupriavidus sp. strain CNP-8. | Min, J., et al. 2018. AMB Express. 8: 43. PMID: 29560541
- Estimation of the Toxicity of Different Substituted Aromatic Compounds to the Aquatic Ciliate Tetrahymena pyriformis by QSAR Approach. | Luan, F., et al. 2018. Molecules. 23: PMID: 29695132
- Characterization of a Pyrethroid-Degrading Pseudomonas fulva Strain P31 and Biochemical Degradation Pathway of D-Phenothrin. | Yang, J., et al. 2018. Front Microbiol. 9: 1003. PMID: 29867894
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Chloro-3-nitrophenol, 5 g | sc-238810 | 5 g | $37.00 |
