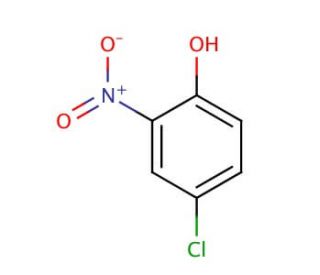
Molecular structure of 4-Chloro-2-nitrophenol, CAS Number: 89-64-5
4-Chloro-2-nitrophenol (CAS 89-64-5)
Alternate Names:
2-Nitro-4-chlorophenol
CAS Number:
89-64-5
Molecular Weight:
173.55
Molecular Formula:
C6H4ClNO3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Chloro-2-nitrophenol is a versatile organic compound that finds extensive utility in the synthesis of diverse organic compounds. It serves as both a reagent and a catalyst in organic synthesis and reactions. The compound is in the synthesis of dyes, and various other organic compounds. 4-Chloro-2-nitrophenol is used as a fluorescent dye in fluorescence microscopy and plays a role in the detection of proteins and other biological molecules. Its electron-accepting properties are well-documented, enabling it to donate electrons to other molecules. 4-Chloro-2-nitrophenol exhibits the ability to form complexes with metal ions, thereby facilitating catalytic reactions that leverage these complexes.
4-Chloro-2-nitrophenol (CAS 89-64-5) References
- Coupled reductive and oxidative degradation of 4-chloro-2-nitrophenol by a co-immobilized mixed culture system. | Beunink, J. and Rehm, HJ. 1990. Appl Microbiol Biotechnol. 34: 108-15. PMID: 1366971
- Degradation mechanisms of 4-chlorophenol in a novel gas-liquid hybrid discharge reactor by pulsed high voltage system with oxygen or nitrogen bubbling. | Zhang, Y., et al. 2007. Chemosphere. 67: 702-11. PMID: 17169402
- Comparison of various advanced oxidation processes for the degradation of 4-chloro-2 nitrophenol. | Saritha, P., et al. 2007. J Hazard Mater. 149: 609-14. PMID: 17703880
- Thermophilic degradation of phenolic compounds in lab scale hybrid up flow anaerobic sludge blanket reactors. | Sreekanth, D., et al. 2009. J Hazard Mater. 164: 1532-9. PMID: 18986764
- Biotransformation of 4-chloro-2-nitrophenol into 5-chloro-2-methylbenzoxazole by a marine Bacillus sp. strain MW-1. | Arora, PK. and Jain, RK. 2012. Biodegradation. 23: 325-31. PMID: 21892663
- Quick photo-Fenton degradation of phenolic compounds by Cu/Al2O3-MCM-41 under visible light irradiation: small particle size, stabilization of copper, easy reducibility of Cu and visible light active material. | Pradhan, AC., et al. 2013. Dalton Trans. 42: 558-66. PMID: 23090390
- Metabolism of 4-chloro-2-nitrophenol in a gram-positive bacterium, Exiguobacterium sp. PMA. | Arora, PK., et al. 2012. Microb Cell Fact. 11: 150. PMID: 23171039
- Decolourization of 4-chloro-2-nitrophenol by a soil bacterium, Bacillus subtilis RKJ 700. | Arora, PK. 2012. PLoS One. 7: e52012. PMID: 23251673
- Comparison of 4-chloro-2-nitrophenol adsorption on single-walled and multi-walled carbon nanotubes. | Mehrizad, A., et al. 2012. Iranian J Environ Health Sci Eng. 9: 5. PMID: 23369489
- Biotransformation and chemotaxis of 4-chloro-2-nitrophenol by Pseudomonas sp. JHN. | Arora, PK. and Bae, H. 2014. Microb Cell Fact. 13: 110. PMID: 25112300
- Transformations of chloro and nitro groups during the peroxymonosulfate-based oxidation of 4-chloro-2-nitrophenol. | Zhou, J., et al. 2015. Chemosphere. 134: 446-51. PMID: 26001937
- Recent advances in degradation of chloronitrophenols. | Arora, PK., et al. 2018. Bioresour Technol. 250: 902-909. PMID: 29229201
- Bacilli-Mediated Degradation of Xenobiotic Compounds and Heavy Metals. | Arora, PK. 2020. Front Bioeng Biotechnol. 8: 570307. PMID: 33163478
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Chloro-2-nitrophenol, 250 g | sc-232552 | 250 g | $155.00 |
