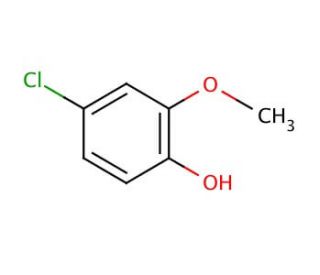
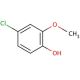
4-Chloro-2-methoxyphenol (CAS 16766-30-6)
QUICK LINKS
4-Chloro-2-methoxyphenol (4-CMP) is a versatile phenolic compound widely employed in the realms of science and technology. Its significance lies in its role as a vital component for synthesizing various organic compounds. The applications of 4-Chloro-2-methoxyphenol extend to serving as an intermediary in drug and dye synthesis, as well as in the production of industrial chemicals. Notably, it finds use as a preservative in food products, a corrosion inhibitor in the petrochemical industry, and a surfactant in cosmetics. Despite the extensive exploration in scientific literature, the precise mechanism of action for 4-Chloro-2-methoxyphenol remains an enigma. Nevertheless, it is believed to influence cellular membranes, heightening their permeability to facilitate the entry of other molecules. Additionally, its interactions with proteins and enzymes are thought to bring about structural and functional alterations. Notably, 4-Chloro-2-methoxyphenol′s antioxidant properties play a role in scavenging free radicals and reducing oxidative stress. 4-Chloro-2-methoxyphenol is an essential compound with diverse applications, ranging from industrial processes to potential medical uses. As the scientific community continues to unravel its mechanisms and explore its various aspects, the prospects for this compound appear promising and worth pursuing.
4-Chloro-2-methoxyphenol (CAS 16766-30-6) References
- Reaction of substituted phenols with thermostable laccase bound to Bacillus subtilis spores. | Hirose, J., et al. 2003. Biotechnol Lett. 25: 1609-12. PMID: 14584915
- Studies in detoxication; the orientation of conjugation in the metabolites of 4-chlorocatechol and 4-chlororesorcinol, with some observations on the fate of (+)-adrenaline, protocatechuic acid and protocatechuic aldehyde in the rabbit. | DODGSON, KS. and WILLIAMS, RT. 1949. Biochem J. 45: 381-6. PMID: 15407243
- Amperometric determination of chloroguaiacol at submicromolar levels after on-line preconcentration with molecularly imprinted polymers. | Tarley, CR., et al. 2006. Talanta. 69: 259-66. PMID: 18970563
- Inhibition of Mycobacterium tuberculosis InhA: Design, synthesis and evaluation of new di-triclosan derivatives. | Armstrong, T., et al. 2020. Bioorg Med Chem. 28: 115744. PMID: 33007556
- Potential of laccase for modification of Eucalyptus globulus wood: a XPS study | , et al. (2014). Wood Science and Technology. volume 48,: pages 151–160.
- Chloroform and chlorophenol production by decarboxylation of natural acids during aqueous chlorination | Richard A. Larson and Arlene L. Rockwell. 1979,. Environ. Sci. Technol. 13, 3,: 325–329.
- Solubility and Infinite Dilution Activity Coefficient for 5-Chlorovanillin and 4-Chloroguaiacol in Water over the Temperature Range 280 to 363 K | Faïçal Larachi, Marc Leroux, Safia Hamoudi, Alain Bernis, and Abdelhamid Sayari. 2000. J. Chem. Eng. Data., 45, 2,: 404–408.
- Permselective Properties of Electropolymerized Guaiacol Derivatives | and Grzegorz Milczarek, Aleksander Ciszewski. April 2003. Electroanalysis. Volume15, Issue5-6: Pages 529-532.
- Optimization of preparation conditions for activated carbon from Prosopis africana seed hulls using response surface methodology | Afidah Abdul Rahim and & Zaharaddeen N. Garba. 2016 -. Desalination and Water Treatment. Volume 57, Issue 38: Pages 17985-17994.
- Electrochemical reduction of 5-chloro-2-(2,4-dichlorophenoxy)phenol (triclosan) in dimethylformamide | KN Knust, MP Foley, MS Mubarak, S Skljarevski… - Journal of …, 2010 - Elsevier. 5 January 2010,. Journal of Electroanalytical Chemistry. Volume 638, Issue 1,: Pages 100-108.
- Diels–Alder reaction of 4-halogenated masked o-benzoquinones with electron-rich dienophiles | SR Surasani, RK Peddinti - Tetrahedron letters, 2011 - Elsevier. 7 September 2011,. Tetrahedron Letters. Volume 52, Issue 36,: Pages 4615-4618.
- Amperometric detection of phenolic compounds with enzyme immobilized in mesoporous silica prepared by electrophoretic deposition | T Shimomura, T Itoh, T Sumiya, T Hanaoka… - Sensors and Actuators B …, 2011 - Elsevier. 20 April 2011,. Sensors and Actuators B: Chemical. Volume 153, Issue 2,: Pages 361-368.
- Molecularly-imprinted solid phase extraction of catechol from aqueous effluents for its selective determination by differential pulse voltammetry | CRT Tarley, LT Kubota - Analytica chimica acta, 2005 - Elsevier. 29 August 2005,. Analytica Chimica Acta. Volume 548, Issues 1–2,: Pages 11-19.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Chloro-2-methoxyphenol, 5 g | sc-226572 | 5 g | $36.00 |
