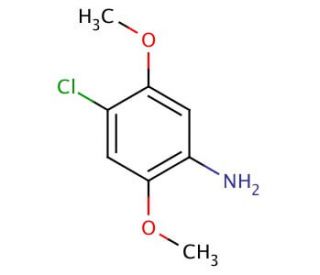
4-Chloro-2,5-dimethoxyaniline (CAS 6358-64-1)
QUICK LINKS
4-Chloro-2,5-dimethoxyaniline functions as a reagent in organic synthesis. It acts as a nucleophilic aromatic substitution reagent, participating in reactions to introduce the 4-chloro-2,5-dimethoxyphenyl group into various organic molecules. 4-Chloro-2,5-Dimethoxyaniline′s mechanism of action involves its ability to undergo substitution reactions with electrophilic aromatic compounds, leading to the formation of new carbon-carbon or carbon-heteroatom bonds. Through its nucleophilic substitution reactions, 4-Chloro-2,5-dimethoxyaniline plays a role in the synthesis of complex organic molecules and the modification of aromatic systems. Its mechanism of action at the molecular level involves the displacement of a leaving group from an aromatic ring, resulting in the formation of a new covalent bond. 4-Chloro-2,5-Dimethoxyaniline′s functional role in organic synthesis involves its participation in the construction of diverse chemical structures through the introduction of the 4-chloro-2,5-dimethoxyphenyl moiety into target molecules.
4-Chloro-2,5-dimethoxyaniline (CAS 6358-64-1) References
- Measurement and correlation of solubility of 4-chloro-2,5-dimethoxynitrobenzene and 4-chloro-2,5-dimethoxyaniline in methanol, ethanol, xylene and toluene | Lin Zhang, Hui Jing, Wen-yu Zhu, Feng-Bao Zhang, Guo-Liang Zhang, Qing Xia. 2015. The Journal of Chemical Thermodynamics. 80: 142-146.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Chloro-2,5-dimethoxyaniline, 100 g | sc-232553 | 100 g | $160.00 |
