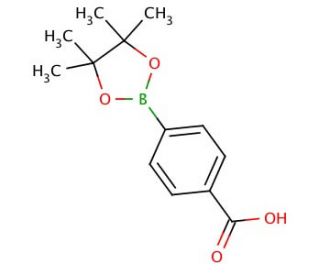
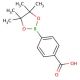
4-Carboxylphenylboronic acid pinacol ester (CAS 180516-87-4)
QUICK LINKS
4-Carboxylphenylboronic acid pinacol ester plays a pivotal role in material science and organic chemistry research due to its stability and reactivity. Its primary application is as a reagent in the Suzuki-Miyaura cross-coupling reactions, which are instrumental in constructing biaryl compounds with precision. The boronic ester moiety specifically interacts with palladium catalysts to facilitate the formation of carbon-carbon bonds, an essential process for synthesizing a variety of complex organic molecules used in OLEDs, liquid crystals, and other electronic materials. Researchers also utilize the carboxyl functional group for post-coupling modifications, such as creating carboxylic acid derivatives, which are key intermediates in the production of dyes, pigments, and polymers with specific light absorption properties. Additionally, this compound is utilized in the creation of boronic acid-based sensors that can detect saccharides and other diol-containing biomolecules, making it useful in the design of diagnostic tools and the study of biological recognition processes.
4-Carboxylphenylboronic acid pinacol ester (CAS 180516-87-4) References
- H2O2-activated oxidative stress amplifier capable of GSH scavenging for enhancing tumor photodynamic therapy. | Liu, Y., et al. 2019. Biomater Sci. 7: 5359-5368. PMID: 31621699
- In Vivo Regenerable Cerium Oxide Nanozyme-Loaded pH/H2O2-Responsive Nanovesicle for Tumor-Targeted Photothermal and Photodynamic Therapies. | Zeng, L., et al. 2021. ACS Appl Mater Interfaces. 13: 233-244. PMID: 33373178
- Synthesis of γ-Oxo-α-amino Acids via Radical Acylation with Carboxylic Acids. | Merkens, K., et al. 2021. J Org Chem. 86: 8448-8456. PMID: 34060842
- Construction of a pH/TGase 'Dual Key'-Responsive Gold Nano-radiosensitizer with Liver Tumor-Targeting Ability. | Zhang, Z., et al. 2021. ACS Biomater Sci Eng. 7: 3434-3445. PMID: 34129333
- The design and synthesis of high efficiency adsorption materials for 1,3-propanediol: physical and chemical structure regulation. | Zheng, K., et al. 2020. RSC Adv. 10: 38085-38096. PMID: 35515184
- A pH, glucose, and dopamine triple-responsive, self-healable adhesive hydrogel formed by phenylborate–catechol complexation | Shan, M., Gong, C., Li, B., & Wu, G. 2017. Polymer Chemistry. 8(19): 2997-3005.
- Boric acid modified macroporous adsorption resin and its adsorption properties for catechol compounds | Liu, B., Liu, J., Huang, D., Wei, J., & Di, D. 2020. Colloids and Surfaces A: Physicochemical and Engineering Aspects. 595: 124674.
- A GSH-depleted platinum (IV) prodrug triggers ferroptotic cell death in breast cancer | Qi, D., Xing, L., Shen, L., Sun, W., Cai, C., Xue, C.,.. & Zhang, Z. 2022. Chinese Chemical Letters. 33(10): 4595-4599.
- Boronic ester bonds crosslinked vitrimer elastomers with mechanical robustness, shape memory, self-healing and recyclability properties | Huang, L., Yang, Y., Niu, Z., Wu, R., Fan, W., Dai, Q.,.. & Bai, C. 2022. Composites Science and Technology. 228: 109621.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Carboxylphenylboronic acid pinacol ester, 5 g | sc-226562 | 5 g | $132.00 |
