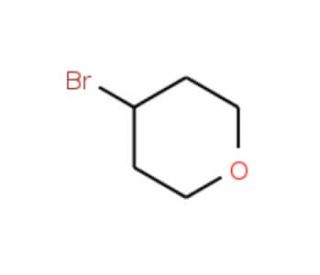
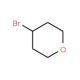
4-Bromotetrahydropyran (CAS 25637-16-5)
QUICK LINKS
4-Bromotetrahydropyran is a synthetic intermediate used in organic chemistry research for the construction of tetrahydropyran rings, which are found in numerous natural products and bioactive molecules. Studies involving 4-Bromotetrahydropyran frequently focus on its role as an electrophile in nucleophilic substitution reactions, where it can be used to introduce tetrahydropyran moieties into larger molecular frameworks. Research on the compound′s reactivity includes its use in coupling reactions and the exploration of its potential as a building block for the assembly of complex ethers. The bromine atom in 4-Bromotetrahydropyran provides a versatile handle for further functionalization, allowing for the introduction of various substituents through palladium-catalyzed cross-coupling reactions, such as Suzuki or Stille couplings. Additionally, the tetrahydropyran ring system of the compound is of interest in studies of ring-opening polymerization, where it can be employed in the synthesis of polymers with defined architectures.
4-Bromotetrahydropyran (CAS 25637-16-5) References
- Titanium(IV)-promoted Mukaiyama aldol-Prins cyclizations. | Patterson, B., et al. 2003. Org Lett. 5: 3163-6. PMID: 12917007
- Synthesis and minisci reactions of organotrifluoroborato building blocks. | Presset, M., et al. 2013. J Org Chem. 78: 4615-9. PMID: 23594305
- Reductive cross-coupling of nonaromatic, heterocyclic bromides with aryl and heteroaryl bromides. | Molander, GA., et al. 2014. J Org Chem. 79: 5771-80. PMID: 24892751
- Discovery of Sulfonamidebenzamides as Selective Apoptotic CHOP Pathway Activators of the Unfolded Protein Response. | Flaherty, DP., et al. 2014. ACS Med Chem Lett. 5: 1278-1283. PMID: 25530830
- Silyl Radical Activation of Alkyl Halides in Metallaphotoredox Catalysis: A Unique Pathway for Cross-Electrophile Coupling. | Zhang, P., et al. 2016. J Am Chem Soc. 138: 8084-7. PMID: 27263662
- New ligands for nickel catalysis from diverse pharmaceutical heterocycle libraries. | Hansen, EC., et al. 2016. Nat Chem. 8: 1126-1130. PMID: 27874864
- Metallaphotoredox-Catalyzed Cross-Electrophile Csp3-Csp3 Coupling of Aliphatic Bromides. | Smith, RT., et al. 2018. J Am Chem Soc. 140: 17433-17438. PMID: 30516995
- The Role Ionic Liquid [BMIM][PF6] in One-Pot Synthesis of Tetrahydropyran Rings through Tandem Barbier-Prins Reaction. | Batista, PK., et al. 2019. Molecules. 24: PMID: 31159274
- Multifunctional Building Blocks Compatible with Photoredox-Mediated Alkylation for DNA-Encoded Library Synthesis. | Badir, SO., et al. 2020. Org Lett. 22: 1046-1051. PMID: 31940210
- EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2019 update. | Smolen, JS., et al. 2020. Ann Rheum Dis. 79: 685-699. PMID: 31969328
- Rapid Optimization of Photoredox Reactions for Continuous-Flow Systems Using Microscale Batch Technology. | González-Esguevillas, M., et al. 2021. ACS Cent Sci. 7: 1126-1134. PMID: 34345665
- Technological Innovations in Photochemistry for Organic Synthesis: Flow Chemistry, High-Throughput Experimentation, Scale-up, and Photoelectrochemistry. | Buglioni, L., et al. 2022. Chem Rev. 122: 2752-2906. PMID: 34375082
- Cathodic Carbonyl Alkylation of Aryl Ketones or Aldehydes with Unactivated Alkyl Halides. | Wu, H., et al. 2022. Org Lett. 24: 9342-9347. PMID: 36484503
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Bromotetrahydropyran, 1 g | sc-261862 | 1 g | $60.00 | |||
4-Bromotetrahydropyran, 5 g | sc-261862A | 5 g | $220.00 |
