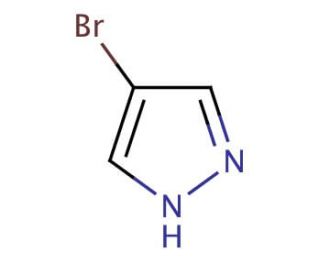
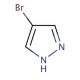
4-Bromopyrazole (CAS 2075-45-8)
QUICK LINKS
4-Bromopyrazole, an organic compound from the pyrazole family, emerges as a fascinating subject of extensive research. Pyrazoles, a group of heterocyclic compounds featuring three carbon atoms and two nitrogen atoms in a five-membered ring, provide a foundation for its classification. The distinctive properties of 4-Bromopyrazole, such as its capacity to form complexes with transition metals and its potential as a ligand in synthetic organic chemistry, have captivated scientific interest. In the realm of scientific research, 4-Bromopyrazole has garnered attention for its versatile applications. Notably, it serves as a valuable biological probe and a catalyst in biochemical reactions. As a biological probe, it aids in the investigation of enzyme activity, protein structure, and the interactions between proteins and small molecules. Furthermore, in biochemical reactions, 4-Bromopyrazole acts as a catalyst, facilitating the synthesis of peptides and engaging in transition metal catalysis. The mechanism of action for 4-Bromopyrazole varies depending on its application. As a biological probe, it selectively binds to target enzymes or proteins, thereby inducing a conformational change within the target molecule. This conformational change serves as a means to study the structure and functionality of the target. In the context of biochemical reactions, 4-Bromopyrazole serves as an activator for substrates, enabling the formation of new chemical bonds.
4-Bromopyrazole (CAS 2075-45-8) References
- Palladium(II) complexes of pyrazolated thio/selenoethers: syntheses, structures, single source precursors of Pd4Se and PdSe nano-particles and potential for catalyzing Suzuki-Miyaura coupling. | Sharma, KN., et al. 2013. Dalton Trans. 42: 3908-18. PMID: 23329305
- Rapid experimental SAD phasing and hot-spot identification with halogenated fragments. | Bauman, JD., et al. 2016. IUCrJ. 3: 51-60. PMID: 26870381
- Halogen bonding properties of 4-iodopyrazole and 4-bromopyrazole explored by rotational spectroscopy and ab initio calculations. | Cooper, GA., et al. 2017. J Chem Phys. 147: 214303. PMID: 29221380
- C-H Imidation and Dual C-H Bond Aminobromination of Five-Membered Heterocycles. | Sun, K., et al. 2020. J Org Chem. 85: 1001-1008. PMID: 31872767
- Mutagenicity study on pyrazole, seven pyrazole derivatives, and two nitroimidazoles with the L-arabinose resistance test of Salmonella typhimurium. | Alejandre-Durán, E., et al. 1986. Environ Mutagen. 8: 611-9. PMID: 3525137
- Effects of pyrazole, 4-bromopyrazole and 4-methylpyrazole on mitochondrial function. | Cederbaum, AI. and Rubin, E. 1974. Biochem Pharmacol. 23: 203-13. PMID: 4360345
- Effect of pyrazole, 4-methylpyrazole, 4-bromopyrazole and 4-iodopyrazole on brain noradrenaline levels of mice and rats. | MacDonald, E. 1976. Acta Pharmacol Toxicol (Copenh). 39: 513-24. PMID: 990035
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Bromopyrazole, 1 g | sc-254636 | 1 g | $55.00 |
