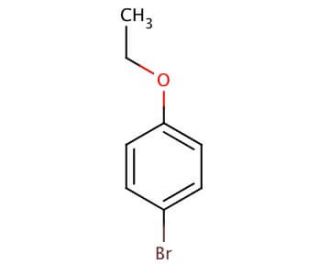
4-Bromophenetole (CAS 588-96-5)
QUICK LINKS
4-Bromophenetole is a chemical compound that functions as a methylation reagent in organic synthesis. It acts as an electrophile, participating in nucleophilic aromatic substitution reactions to introduce a methoxy group into various aromatic compounds. 4-Bromophenetole′s mode of action involves the substitution of a hydrogen atom on the aromatic ring with a methoxy group, resulting in the formation of a new carbon-oxygen bond. This reaction occurs under specific reaction conditions, allowing for the selective introduction of the methoxy group at desired positions on the aromatic ring. In the and development, 4-Bromophenetole serves for the modification of aromatic compounds, enabling the synthesis of diverse organic molecules with tailored properties. Its precise mode of action at the molecular level makes it a useful reagent for the creation of novel chemical structures with potential applications in various fields.
4-Bromophenetole (CAS 588-96-5) References
- Draize rabbit eye test compatibility with eye irritation thresholds in humans: a quantitative structure-activity relationship analysis. | Abraham, MH., et al. 2003. Toxicol Sci. 76: 384-91. PMID: 14514959
- Simple stochastic fingerprints towards mathematical modeling in biology and medicine. 3. Ocular irritability classification model. | Cruz-Monteagudo, M., et al. 2006. Bull Math Biol. 68: 1555-72. PMID: 16865609
- Structure-function relationships of estrogenic triphenylethylenes related to endoxifen and 4-hydroxytamoxifen. | Maximov, PY., et al. 2010. J Med Chem. 53: 3273-83. PMID: 20334368
- Practical applications of QSAR to in vitro toxicology illustrated by consideration of eye irritation. | Chamberlain, M. and Barratt, MD. 1995. Toxicol In Vitro. 9: 543-7. PMID: 20650126
- QSARS for the eye irritation potential of neutral organic chemicals. | Barratt, MD. 1997. Toxicol In Vitro. 11: 1-8. PMID: 20654291
- Draize Eye Scores and Eye Irritation Thresholds in Man Combined into one Quantitative Structure-Activity Relationship. | H Abraham, M., et al. 1998. Toxicol In Vitro. 12: 403-8. PMID: 20654423
- A quantitative structure-activity relationship (QSAR) investigation of a Draize eye irritation database. | Cronin, MT., et al. 1994. Toxicol In Vitro. 8: 21-8. PMID: 20692885
- Novel thiophenyl C-aryl glucoside SGLT2 inhibitors as potential antidiabetic agents. | Lee, SH., et al. 2011. Bioorg Med Chem. 19: 5813-32. PMID: 21906953
- Evaluation of Sydnone-Based Analogues of Combretastatin A-4 Phosphate (CA4P) as Vascular Disrupting Agents for Use in Cancer Therapy. | Brown, AW., et al. 2018. ChemMedChem. 13: 2618-2626. PMID: 30281922
- Development of a defined approach for eye irritation or serious eye damage for liquids, neat and in dilution, based on cosmetics Europe analysis of in vitro STE and BCOP test methods. | Alépée, N., et al. 2019. Toxicol In Vitro. 57: 154-163. PMID: 30817952
- The EyeIRR-IS assay: Development and evaluation of an in vitro assay to measure the eye irritation sub-categorization of liquid chemicals. | Cottrez, F., et al. 2021. Toxicol In Vitro. 71: 105072. PMID: 33358762
- The use of cluster significance analysis to identify asymmetric QSAR data sets in toxicology. An example with eye irritation data. | Cronin, MT. 1996. SAR QSAR Environ Res. 5: 167-75. PMID: 9114513
- Integration of QSAR and in vitro toxicology. | Barratt, MD. 1998. Environ Health Perspect. 106 Suppl 2: 459-65. PMID: 9599692
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Bromophenetole, 100 g | sc-232511 | 100 g | $79.00 |
