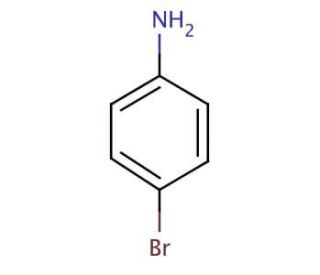
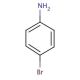
4-Bromoaniline (CAS 106-40-1)
QUICK LINKS
4-Bromoaniline is a chemical compound that functions as a building block in organic synthesis. It acts as a precursor in the production of various dyes, and agrochemicals. Its mechanism of action involves participating in nucleophilic aromatic substitution reactions, where the bromine atom is substituted with other functional groups to create new compounds. 4-Bromoaniline plays a role in the modification of aromatic compounds, allowing for the introduction of specific functional groups at the para position. In experimental applications, 4-Bromoaniline is utilized to facilitate the synthesis of complex organic molecules by serving as a starting material for the construction of diverse chemical structures. Its molecular properties enable it to undergo various chemical transformations, making it a versatile component in the development of novel compounds.
4-Bromoaniline (CAS 106-40-1) References
- High-performance liquid chromatography and inductively coupled plasma mass spectrometry (HPLC-ICP-MS) for the analysis of xenobiotic metabolites in rat urine: application to the metabolites of 4-bromoaniline. | Nicholson, JK., et al. 2000. Analyst. 125: 235-6. PMID: 10820889
- Identification of the urinary metabolites of 4-bromoaniline and 4-bromo-[carbonyl-13C]-acetanilide in rat. | Scarfe, GB., et al. 2002. Xenobiotica. 32: 325-37. PMID: 12028665
- Characterisation of putative pentose-containing conjugates as minor metabolites of 4-bromoaniline present in the urine of rats following intraperitoneal administration. | Major, H., et al. 2003. Rapid Commun Mass Spectrom. 17: 76-80. PMID: 12478557
- Detection of mono- and di-hexoses as metabolites of 4-bromoaniline using HPLC-TOF-MS/MS. | Major, H., et al. 2003. Xenobiotica. 33: 855-69. PMID: 12936705
- Potential of neurotoxicity after a single oral dose of 4-bromo-, 4-chloro-, 4-fluoro- or 4-iodoaniline in rats. | Okazaki, Y., et al. 2003. J Appl Toxicol. 23: 315-22. PMID: 12975770
- Effects of dissolved organic carbon on sorption of 3,4-dichloroaniline and 4-bromoaniline in a calcareous soil. | González-Pradas, E., et al. 2005. Chemosphere. 59: 721-8. PMID: 15792670
- Proton-transfer compounds of 8-hydroxy-7-iodoquinoline-5-sulfonic acid (ferron) with 4-chloroaniline and 4-bromoaniline. | Smith, G., et al. 2007. Acta Crystallogr C. 63: o405-7. PMID: 17609572
- Mechanism of inhibition of myeloperoxidase by anti-inflammatory drugs. | Kettle, AJ. and Winterbourn, CC. 1991. Biochem Pharmacol. 41: 1485-92. PMID: 1850278
- The metabolism of 4-bromoaniline in the bile-cannulated rat: application of ICPMS ((79/81)Br), HPLC-ICPMS & HPLC-oaTOFMS. | Duckett, C., et al. 2015. Xenobiotica. 45: 672-80. PMID: 25837688
- Studies on Oligomer Metal Complexes Derived from Bisamic Acid of Pyromellitic Dianhydride and 4-Bromoaniline. | Patel, YS. 2014. Int Sch Res Notices. 2014: 516274. PMID: 27379295
- N-([1,1'-biaryl]-4-yl)-1-naphthamide-based scaffolds synthesis, their cheminformatics analyses, and screening as bacterial biofilm inhibitor. | Ejaz, S., et al. 2022. J Basic Microbiol. 62: 1143-1155. PMID: 34724237
- Bright Luminescent Carbon Dots for Multifunctional Selective Sensing and Imaging Applications in Living Cells. | Thirumalaivasan, N. and Wu, SP. 2020. ACS Appl Bio Mater. 3: 6439-6446. PMID: 35021775
- Synthesis of Functionalized N-(4-Bromophenyl)furan-2-carboxamides via Suzuki-Miyaura Cross-Coupling: Anti-Bacterial Activities against Clinically Isolated Drug Resistant A. baumannii, K. pneumoniae, E. cloacae and MRSA and Its Validation via a Computational Approach. | Siddiqa, A., et al. 2022. Pharmaceuticals (Basel). 15: PMID: 35890140
- Microbial mineralization of ring-substituted anilines through an ortho-cleavage pathway. | Zeyer, J., et al. 1985. Appl Environ Microbiol. 50: 447-53. PMID: 4051488
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Bromoaniline, 5 g | sc-256720 | 5 g | $17.00 | |||
4-Bromoaniline, 100 g | sc-256720A | 100 g | $65.00 |
