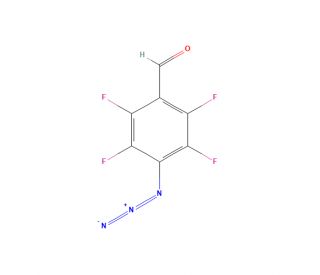
4-Azidotetrafluorobenzaldehyde (CAS 120384-18-1)
QUICK LINKS
4-Azidotetrafluorobenzaldehyde is a specialized chemical compound that features prominently in material science and synthetic organic chemistry research due to its unique functional groups: an azide and a tetrafluorobenzaldehyde. This combination offers multiple reactive sites, making it a valuable tool for constructing complex molecular architectures. The azide group in 4-Azidotetrafluorobenzaldehyde is highly reactive and can undergo various types of reactions, including the well-known click chemistry reactions (specifically, azide-alkyne cycloaddition). This reactivity is exploited in the synthesis of diverse heterocyclic compounds and in the modification of surfaces or the creation of polymer networks, where the azide serves as a crucial linking functionality. Meanwhile, the tetrafluorobenzaldehyde moiety impacts the electronic properties of molecules, increasing their electrophilic character. This can lead to interesting interactions in systems where electron density plays a critical role, such as in sensors or organic electronics. The fluorine atoms also enhance the stability and rigidity of the molecules, which is beneficial in applications requiring high thermal and chemical resistance. In research contexts, 4-Azidotetrafluorobenzaldehyde is used to study reaction mechanisms and kinetics, especially where azide functionalities are involved. Its application in developing novel synthetic pathways and materials underscores its significance in advancing the fields of chemistry and materials science, offering insights into the construction of molecular systems with tailored properties.
4-Azidotetrafluorobenzaldehyde (CAS 120384-18-1) References
- Unexpected formal [1+3] cycloadditions between azides and alpha-zirconated phosphanes: a route to unprecedented phosphazide and iminophosphorane complexes. | Cadierno, V., et al. 2000. Chemistry. 6: 345-52. PMID: 11931115
- Ginkgolide derivatives for photolabeling studies: preparation and pharmacological evaluation. | Strømgaard, K., et al. 2002. J Med Chem. 45: 4038-46. PMID: 12190325
- Photochemical functionalization of polymer surfaces for microfabricated devices. | Mecomber, JS., et al. 2008. Langmuir. 24: 3645-53. PMID: 18294015
- Photoactivatable phospholipids bearing tetrafluorophenylazido chromophores exhibit unprecedented protonation-state-dependent 19F NMR signals. | Xia, Y., et al. 2011. Org Lett. 13: 4248-51. PMID: 21766799
- A bola-phospholipid bearing tetrafluorophenylazido chromophore as a promising lipid probe for biomembrane photolabeling studies. | Xia, Y., et al. 2013. Org Biomol Chem. 11: 5000-5. PMID: 23783551
- Multiplex Microarrays in 96-Well Plates Photoactivated with 4-Azidotetrafluorobenzaldehyde for the Identification and Quantification of β-Lactamase Genes and Their RNA Transcripts. | Ulyashova, MM., et al. 2023. Curr Issues Mol Biol. 46: 53-66. PMID: 38275665
- Photoactivatable 2-[(4'-azido)tetrafluorophenyl]-5-tert-butyl-1,3-dithiane bissulfone and related compounds as candidate irreversible probes for the GABA-gated chloride channels | Isabelle Kapfer, Jon E. Hawkinson, John E. Casida, and Maurice P. Goeldner. 1994. Journal of Medicinal Chemistry. 37: 133–140.
- Chemistry of Bifunctional Photoprobes.1 3. Correlation between the Efficiency of CH Insertion by Photolabile Chelating Agents and Lifetimes of Singlet Nitrenes by Flash Photolysis: First Example of Photochemical Attachment of 99mTc−Complex with Human Serum Albumin | Raghoottama S. Pandurangi, Przemyslaw Lusiak, Robert R. Kuntz, Wynn A. Volkert, Jacek Rogowski, and Matthew S. Platz. 1998. The Journal of Organic Chemistry. 63: 9019–9030.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Azidotetrafluorobenzaldehyde, 250 mg | sc-503200 | 250 mg | $380.00 |
