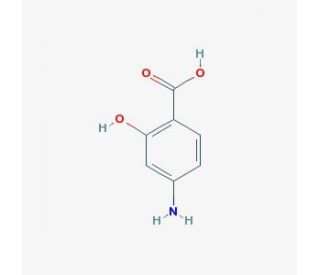
4-Aminosalicylic acid (CAS 65-49-6)
QUICK LINKS
4-Aminosalicylic acid, also known as para-aminosalicylic acid or PAS, is a compound that has been historically significant in biochemical research, particularly in the study of its anti-inflammatory properties. The primary mechanism of action of 4-aminosalicylic acid involves the inhibition of the synthesis of inflammatory mediators, largely through its interference with the metabolism and functions of arachidonic acid, a precursor to prostaglandins and leukotrienes that are pivotal in the inflammatory process. It achieves this by hindering the activity of enzymes such as cyclooxygenase and lipoxygenase, thereby reducing the production of these inflammatory compounds. In research contexts, 4-aminosalicylic acid has been extensively used to explore cellular pathways of inflammation and to develop an understanding of how inflammation can be controlled at a molecular level. Additionally, its role in modulating the production of free radicals and reactive oxygen species during inflammatory responses has been a focus, helping to explain the oxidative stress component of various pathological conditions. These studies contribute significantly to the fundamental understanding of inflammation and offer insights into potential mechanisms for managing diseases characterized by excessive inflammatory responses, thereby advancing knowledge in cell biology and biochemistry.
4-Aminosalicylic acid (CAS 65-49-6) References
- Colonic delivery of 4-aminosalicylic acid using amylose-ethylcellulose-coated hydroxypropylmethylcellulose capsules. | Tuleu, C., et al. 2002. Aliment Pharmacol Ther. 16: 1771-9. PMID: 12269970
- Tolerance of 4-aminosalicylic acid enemas in patients with inflammatory bowel disease and 5-aminosalicylic-induced acute pancreatitis. | Daniel, F., et al. 2004. Inflamm Bowel Dis. 10: 258-60. PMID: 15290921
- Colon-specific mutual amide prodrugs of 4-aminosalicylic acid for their mitigating effect on experimental colitis in rats. | Dhaneshwar, SS., et al. 2009. Eur J Med Chem. 44: 131-42. PMID: 18472188
- Colon-specific prodrugs of 4-aminosalicylic acid for inflammatory bowel disease. | Dhaneshwar, SS. 2014. World J Gastroenterol. 20: 3564-71. PMID: 24707139
- Polymorphism in sulfadimidine/4-aminosalicylic acid cocrystals: solid-state characterization and physicochemical properties. | Grossjohann, C., et al. 2015. J Pharm Sci. 104: 1385-98. PMID: 25605031
- Experience with topical administration of 4-aminosalicylic acid in ulcerative colitis. | Nagy, F., et al. 1989. Dis Colon Rectum. 32: 134-7. PMID: 2563344
- Synthesis, colon-targeted studies and pharmacological evaluation of an anti-ulcerative colitis drug 4-Aminosalicylic acid-β-O-glucoside. | Li, F., et al. 2016. Eur J Med Chem. 108: 486-494. PMID: 26717200
- Modelling and shadowgraph imaging of cocrystal dissolution and assessment of in vitro antimicrobial activity for sulfadimidine/4-aminosalicylic acid cocrystals. | Serrano, DR., et al. 2016. Eur J Pharm Sci. 89: 125-36. PMID: 27131605
- Drug-drug cocrystals of antituberculous 4-aminosalicylic acid: Screening, crystal structures, thermochemical and solubility studies. | Drozd, KV., et al. 2017. Eur J Pharm Sci. 99: 228-239. PMID: 28011126
- Synthesis, biological evaluation and molecular docking study of some new 4-aminosalicylic acid derivatives as anti-inflammatory and antimycobacterial agents. | Qahtan, MQM., et al. 2023. Bioorg Chem. 132: 106344. PMID: 36669356
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Aminosalicylic acid, 10 g | sc-277101 | 10 g | $32.00 |
