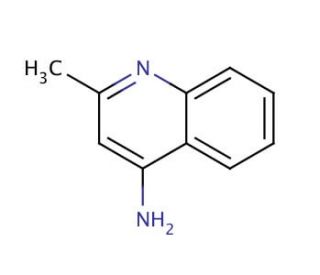
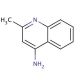
4-Aminoquinaldine (CAS 6628-04-2)
QUICK LINKS
4-Aminoquinaldine (4-AQ), an organic compound derived from quinaldine, has garnered considerable attention for its potential applications across diverse scientific research fields. This compound holds immense significance as an intermediate in synthesizing various organic compounds. In scientific research, 4-Aminoquinaldine has found utility in several applications. In medicinal chemistry, it serves as a foundational component for synthesizing a wide array of organic compounds. Additionally, in the realm of biochemistry, it has facilitated investigations into the influence of specific enzymes on 4-Aminoquinaldine metabolism. While the precise mechanism of action of 4-Aminoquinaldine remains incompletely understood, it is believed to function as an inhibitor of certain enzymes. Specifically, it is thought to impede the activity of enzymes involved in quinaldine metabolism.
4-Aminoquinaldine (CAS 6628-04-2) References
- Interaction of Clostridium botulinum C2 toxin with lipid bilayer membranes and Vero cells: inhibition of channel function by chloroquine and related compounds in vitro and intoxification in vivo. | Bachmeyer, C., et al. 2001. FASEB J. 15: 1658-60. PMID: 11427518
- The antibacterial activity of new derivatives of 4-aminoquinoline and 4-aminoquinaldine. | CALDWELL, D., et al. 1961. J Pharm Pharmacol. 13: 554-64. PMID: 13689922
- CHEMOTHERAPEUTIC PROPERTIES OF SOME NEW QUATERNARY AMMONIUM COMPOUNDS; THEIR CESTICIDAL ACTION AGAINST HYMENOLEPIS NANA. | BHATTACHARYA, BK. and SEN, AB. 1965. Br J Pharmacol Chemother. 24: 240-4. PMID: 14302357
- Modulation of Ca(2+)-dependent and Ca(2+)-independent miniature endplate potentials by phorbol ester and adenosine in frog. | Searl, TJ. and Silinsky, EM. 2005. Br J Pharmacol. 145: 954-62. PMID: 15880138
- Parallel synthesis of diarylureas and their evaluation as inhibitors of insulin-like growth factor receptor. | Anderson, MO., et al. 2006. J Comb Chem. 8: 784-90. PMID: 16961415
- Synthesis of aryl-heteroaryl ureas (AHUs) based on 4-aminoquinoline and their evaluation against the insulin-like growth factor receptor (IGF-1R). | Engen, W., et al. 2010. Bioorg Med Chem. 18: 5995-6005. PMID: 20643554
- Evaluation of Diarylureas for Activity Against Plasmodium falciparum. | Zhang, Y., et al. 2010. ACS Med Chem Lett. 1: 460-465. PMID: 21243104
- Discovery of safe and orally effective 4-aminoquinaldine analogues as apoptotic inducers with activity against experimental visceral leishmaniasis. | Palit, P., et al. 2012. Antimicrob Agents Chemother. 56: 432-45. PMID: 22024817
- Synthesis and biological evaluation of new sulfonamide derivatives as potential anti-Trypanosoma cruzi agents. | Bocanegra-Garcia, V., et al. 2012. Med Chem. 8: 1039-44. PMID: 22762161
- Solid state forms of 4-aminoquinaldine - From void structures with and without solvent inclusion to close packing. | Braun, DE., et al. 2015. CrystEngComm. 17: 2504-2516. PMID: 26726294
- Can computed crystal energy landscapes help understand pharmaceutical solids? | Price, SL., et al. 2016. Chem Commun (Camb). 52: 7065-77. PMID: 27067116
- Ethanol and water sorption into stratum corneum and model systems. | Berner, B., et al. 1989. J Pharm Sci. 78: 472-6. PMID: 2760822
- 4-Aminoquinaldine monohydrate polymorphism: Prediction and impurity aided discovery of a difficult to access stable form. | Braun, DE., et al. 2016. CrystEngComm. 18: 4053-4067. PMID: 28649176
- Effects of local anesthetics and related compounds on the endocytosis and catabolism of asialo-glycoproteins in isolated hepatocytes. | Tolleshaug, H., et al. 1982. Biochim Biophys Acta. 714: 114-21. PMID: 7055602
- The effect of ionic strength on DNA-ligand unwinding angles for acridine and quinoline derivatives. | Jones, RL., et al. 1980. Nucleic Acids Res. 8: 1613-24. PMID: 7191995
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Aminoquinaldine, 5 g | sc-238773 | 5 g | $82.00 |
