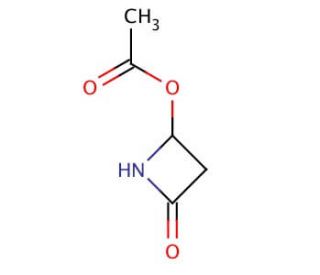
Molecular structure of 4-Acetoxy-2-azetidinone, CAS Number: 28562-53-0
4-Acetoxy-2-azetidinone (CAS 28562-53-0)
CAS Number:
28562-53-0
Molecular Weight:
129.11
Molecular Formula:
C5H7NO3
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
4-Acetoxy-2-azetidinone serves as a model in the synthesis of β-amino acids. A process of allylation at its α position produces an enolate, which can then react with a nitrogen nucleophile to produce an α,β unsaturated carbonyl group. The versatile carbonyl unit can be either hydrolyzed to yield a hydroxyl group or chlorinated to produce a chloride. It has been used in asymmetric synthesis as a chiral auxiliary. Additionally, it plays a part in the synthesis of derivatized cyclopentenes, exhibiting high regio- and diastereoselectivity. It has also been used as a heterocyclic synthon for antibiotic and anti-inflammatory agents.
4-Acetoxy-2-azetidinone (CAS 28562-53-0) References
- The indium-mediated selective introduction of allenyl and propargyl groups at the C4-position of 2-azetidinones and the AuCl3-catalyzed cyclization of 4-allenyl-2-azetidinones. | Lee, PH., et al. 2005. Angew Chem Int Ed Engl. 44: 1840-3. PMID: 15714454
- Indium-mediated 1,2,4,5-hexatetraen-3-ylation of 4-acetoxy-2-azetidinones and their applications to the Diels-Alder reactions for the synthesis of 2-azetidinone derivatives. | Yu, H. and Lee, PH. 2008. J Org Chem. 73: 5183-6. PMID: 18507447
- Pd(0)/InI-mediated allylic additions to 4-acetoxy-2-azetidinone: new route to highly functionalized carbocyclic scaffolds. | Cesario, C. and Miller, MJ. 2009. Org Lett. 11: 1293-5. PMID: 19216521
- Syntheses of carbocyclic uracil polyoxin C analogs: application of Pd(0)/InI-allylation of 4-acetoxy-2-azetidinone. | Cesario, C. and Miller, MJ. 2009. J Org Chem. 74: 5730-3. PMID: 19514728
- Phytochemical Studies, Antioxidant Potential, and Identification of Bioactive Compounds Using GC-MS of the Ethanolic Extract of Luffa cylindrica (L.) Fruit. | S, AS. and Vellapandian, C. 2022. Appl Biochem Biotechnol. 194: 4018-4032. PMID: 35583705
- Reaction of 4-acetoxy-2-azetidinone with tertiary carbanions: Preparation of 4-alkyl- and 4-alkylidene-2-azetidinones | C.W. Greengrass ∗, D.W.T. Hoople. 1981. Tetrahedron Letters. 22: 1161-1164.
- Studies on 1-carbadethiacephems, part II: Reaction of 4-acetoxy-2-azetidinone with aluminium enolates | C.W. Greengrass ∗, M.S. Nobbs. 1981. Tetrahedron Letters. 22: 5339-5340.
- New synthesis of 4-acetoxy-2-azetidinones by use of electrochemical oxidation | Miwako Mori ∗, Katsuji Kagechika, Koh Tohjima, Masakatsu Shibasaki ∗. 1988. Tetrahedron Letters. 29: 1409-1412.
- 2-aza-1,3-dienes as novel precursors for the synthesis of N -unsubstituted β-lactams. A three step synthesis of 4-acetoxy-3-phenoxy-2-azeridinone | Gunda I Georg ∗, Joydeep Kant, Ping He, Ana Maria Ly, Lynn Lampe 1. 1988. Tetrahedron Letters. 29: 2409-2412.
- Reaction of 4-substituted 2-azetidinone with nucleophiles. Existence and reactivity of 1-azetin-4-one | Francisco Gavina, Ana M. Costero, and M. Rosario Andreu, et al. 1990. J. Org. Chem. 55: 434–437.
- Highly stereocontrolled synthesis of the 1β-methylcarbapenem key intermediate by the reformatsky reaction of 3-(2-bromopropionyl)-2-oxazolidone derivatives with a 4-acetoxy-2-azetidinon | Yoshio Ito a 1, Akira Sasaki b, Kastumi Tamoto b, Makoto Sunagawa b, Shiro Terashima ∗ a. 1991. Tetrahedron. 47: 2801-2820.
- Synthesis of 4-allenyl and 4-proparyl-2-azetidinone via Zn-mediated Barbier-type reaction and Pt-catalyzed intramolecular amidation to carbapenem skeletons | Biao Jiang, Hua Tian. 2007. Tetrahedron Letters. 48: 7942-7945.
- Practical synthetic approach to 4-acetoxy-2-azetidinone for the preparation of carbapenem and penem antibiotics | Guo-Bin Zhou, Yue-Qing Guan, He Tang, Yan-Bin Zhao & Li-Rong Yang. 2012. Research on Chemical Intermediates. 38: 251–259.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Acetoxy-2-azetidinone, 1 g | sc-254591 | 1 g | $94.00 |
